Understanding mechanisms driving the stressor-to-phenotype phenomena and controlling these maladaptive responses in the wake of stressor presentation may lead to the development of rational therapeutic approaches to abrogate dysfunction from interactomes to connectomes.

On the cover: Essential to normal cellular function is the interaction among proteins and their organization in well-defined networks. Pillarsetty et al. (pp. 559–573) introduced a positron emission tomography-based assay called PU-PET to visualize pathologic protein-protein interaction networks termed epichaperomes in human cancer patients. They demonstrate epichaperome engagement by the drug candidate PU-H71 in patients’ tumors at single-lesion resolution and show how information derived from PU-PET provides a platform for precision medicine targeting of the aberrant properties of protein networks. Artwork by Samantha Welker and Samantha Gore. [courtesy of Cancer Cell]
Chiosis G, Digwal CS, Trepel JB, Neckers L. Structural and functional complexity of HSP90 in cellular homeostasis and disease. Nat Rev Mol Cell Biol. 2023. doi: 10.1038/s41580-023-00640-9. https://pubmed.ncbi.nlm.nih.gov/37524848/
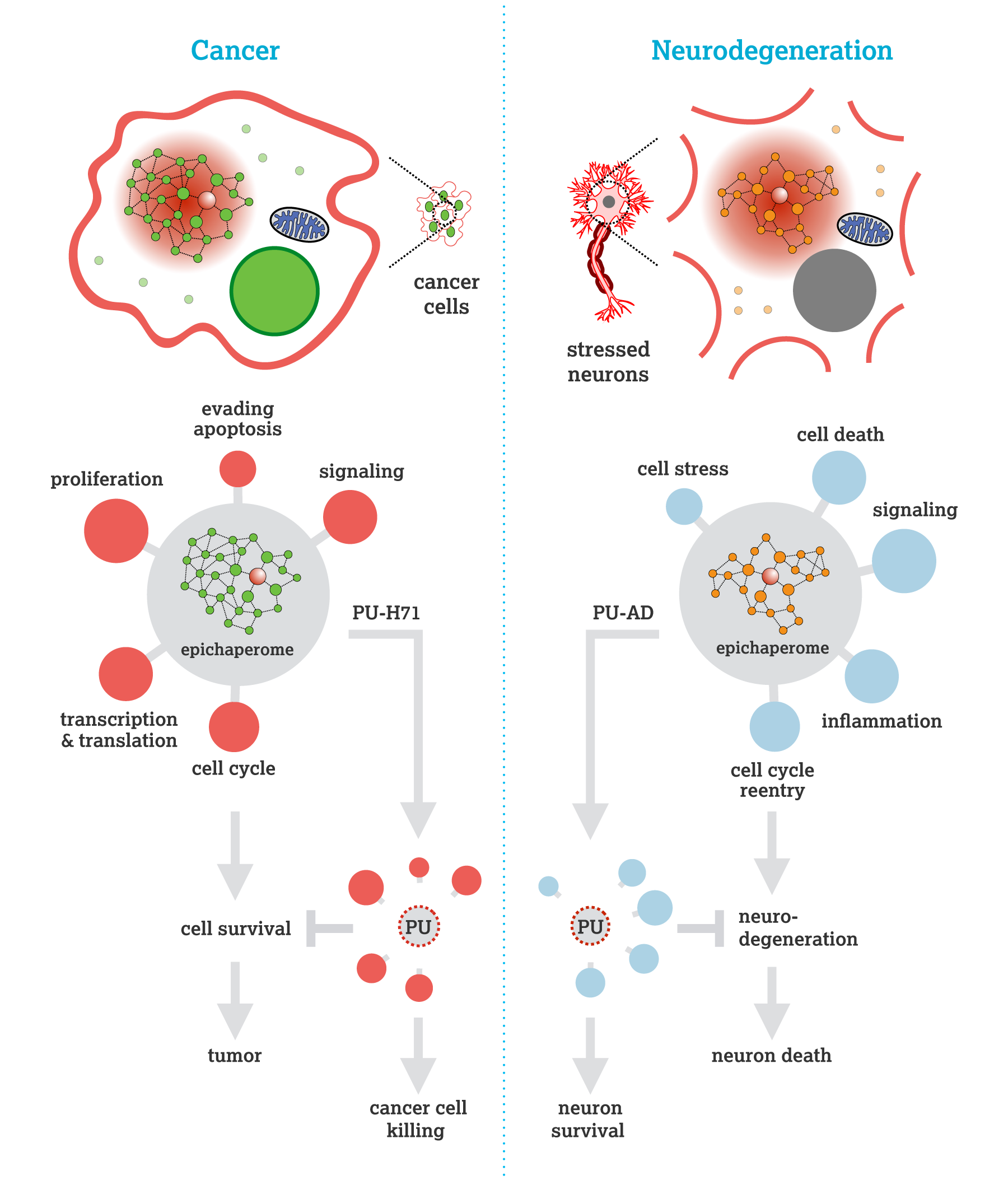
Ginsberg SD, Sharma S, Norton L, Chiosis G. Targeting stressor-induced dysfunctions in protein-protein interaction networks via epichaperomes. Trends Pharmacol Sci. 2023;44(1):20-33. doi: 10.1016/j.tips.2022.10.006. https://pubmed.ncbi.nlm.nih.gov/36414432/
Ginsberg SD, Joshi S, Sharma S, Guzman G, Wang T, Arancio O, Chiosis G. The penalty of stress - Epichaperomes negatively reshaping the brain in neurodegenerative disorders. J Neurochem. 2021;159(6):958-79. https://pubmed.ncbi.nlm.nih.gov/34657288/