Cellular senescence is a stress-response program (Figure 1) that is potentially tumor suppressive. We previously showed that senescence arises through chromatin remodeling that silences proliferative genes while activating genes encoding secreted proteins (1-5). This latter component, termed the senescence-associated secretory phenotype (SASP), has potent effects on the tissue environment that can lead to the immune-mediated targeting of senescent cells (2, 5-7). However, the biological effects of senescent cells are heterogeneous. Whereas we and others have shown that senescence can facilitate wound resolution (8-10), the aberrant persistence of senescent cells in chronically-damaged tissues contributes to cancer progression (11-17) and to a range of non-cancer pathologies (18). Understanding the mechanisms that dictate this diverse biology remains a major goal.
Recent efforts from our laboratory have focused on the roles and regulation of the SASP and its action in immune surveillance of senescent cells (19, 20). Using a well-controlled setting where senescence can be induced in liver tumors by reengaging the p53 tumor suppressor, we showed how remodeling of the surface proteome of senescent cells alters their ability to sense and respond to microenvironmental signals. As one example, senescent cells become hypersensitive to interferon-g and become more effective antigen-presenting cells (21). Together, it appears senescent cells alter, and are affected by, their microenvironment.
Emerging data suggest substantial heterogeneity in the senescence program (19, 22-25) that we plan to systematically characterize using our powerful models, single-cell approaches, and functional perturbations. Beyond identifying better senescence biomarkers, we aim to achieve a mechanistic understanding of factors that dictate senescent cell persistence or clearance (Figure 2).
From a therapeutic standpoint, we are interested in provoking senescence-associated immune surveillance in tumor cells and blunting the deleterious effects of senescent cells in pathologies associated with chronic tissue damage and aging. Hence, we continue to characterize how senescence induction modulates the immune response and are testing new combination strategies based on this understanding. Current studies, in mice, indicate that some drugs that trigger senescence can provoke immune-mediated clearance and/or enhanced response to immune checkpoint blockade (19, 20, 26). A long-term goal is to use our understanding of senescence biology to increase the fraction of cancer patients who benefit from immunotherapy.
There is also substantial interest in ‘senolytic’ drugs that can eliminate senescent cells from damaged tissue. While promising, many current agents are toxic and/or their mechanism of action is unclear. Taking a different approach, we collaborated with Michel Sadelain to produce chimeric antigen receptor (CAR) T cells that target a protein selectively upregulated during senescence. These CAR T cells efficiently eliminate senescent cells from tissues (Figure 3), enhance the activity of senescence-inducing therapy, and ameliorate liver fibrosis (27). More recently we showed that senolytic CAR T cells markedly improved metabolic dysfunction, even when the CAR T cells were delivered prophylactically (28). These studies establish cell-based therapy as a viable strategy to treat senescence-associated pathologies and pave the way to use such therapies for non-cancer diseases. Current studies focus on expanding these efforts and developing second-generation approaches for targeting senescent cells.
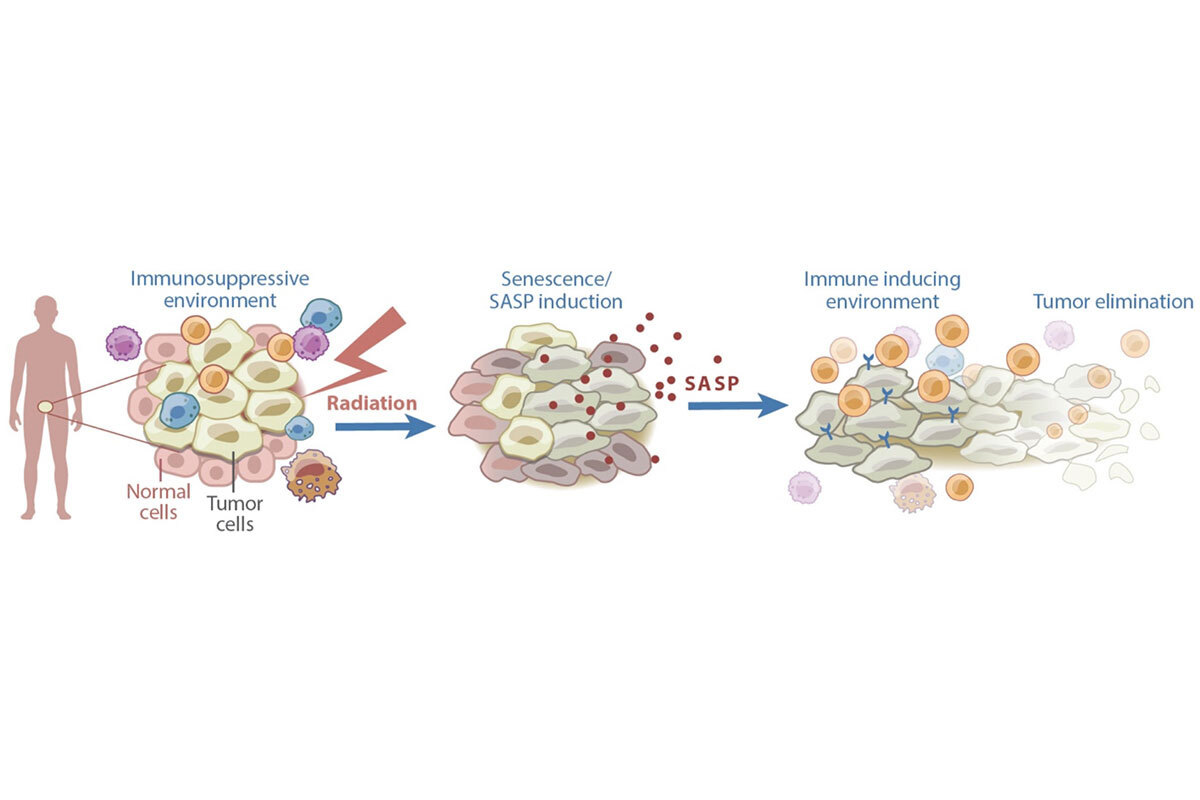
Figure 1. Senescence can contribute to anti-cancer effects of therapy. Certain cancer treatments, including radiation therapy, can induce senescence and the SASP. The SASP subsequently remodels the tumor microenvironment in a manner that induces immune surveillance of the tumor.
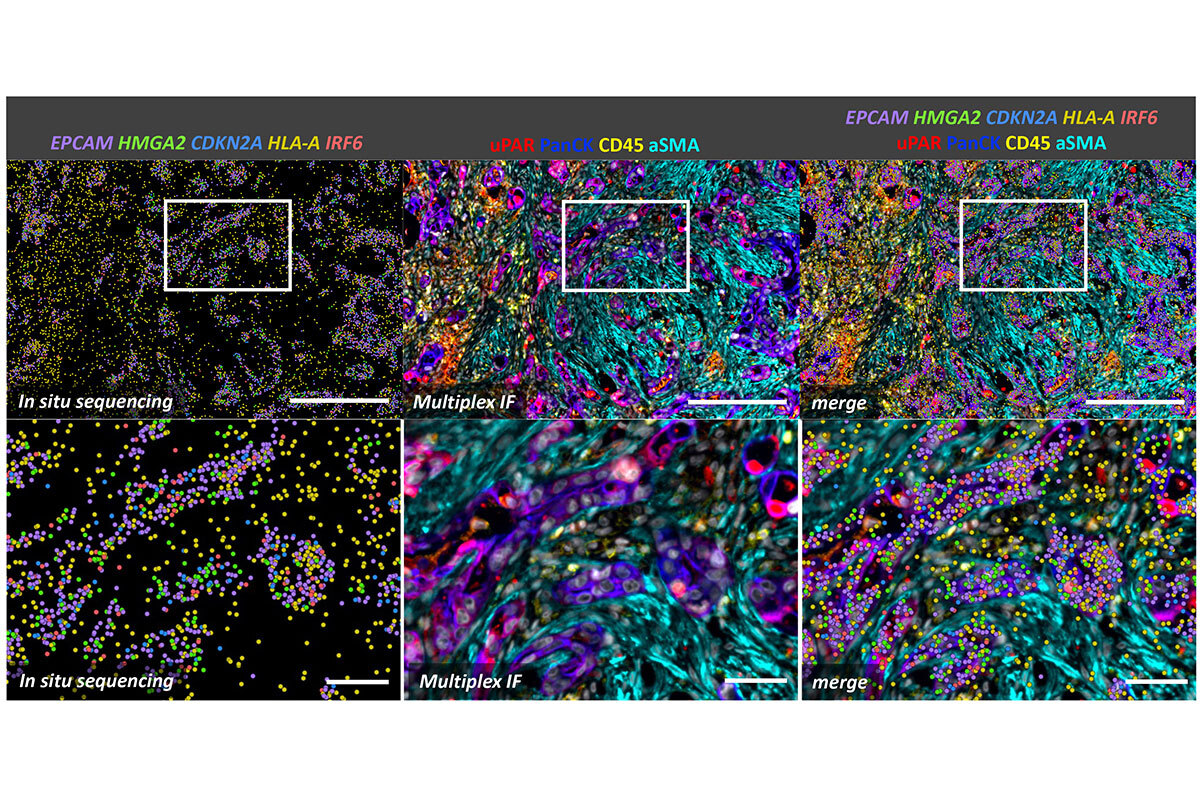
Figure 2. Spatial analysis of senescence at the single-cell level of pancreatic cancer treated with a senescence-inducing chemotherapy. In situ sequencing on the Xenium platform was followed by multiplex immunofluorescence using the CellDive instrument. Senescent cells, characterized by the expression of HMGA2, CDKN2A, and uPAR markers, are enriched among tumor cells (PanCK+ cells). Senescent tumor cells show activation of interferon signaling (IRF6) alongside the expression of MHC I markers (HLA-A), which enhances their susceptibility to immune system recognition. Scale bars: 250 µm in top row and 50 µm in bottom row.
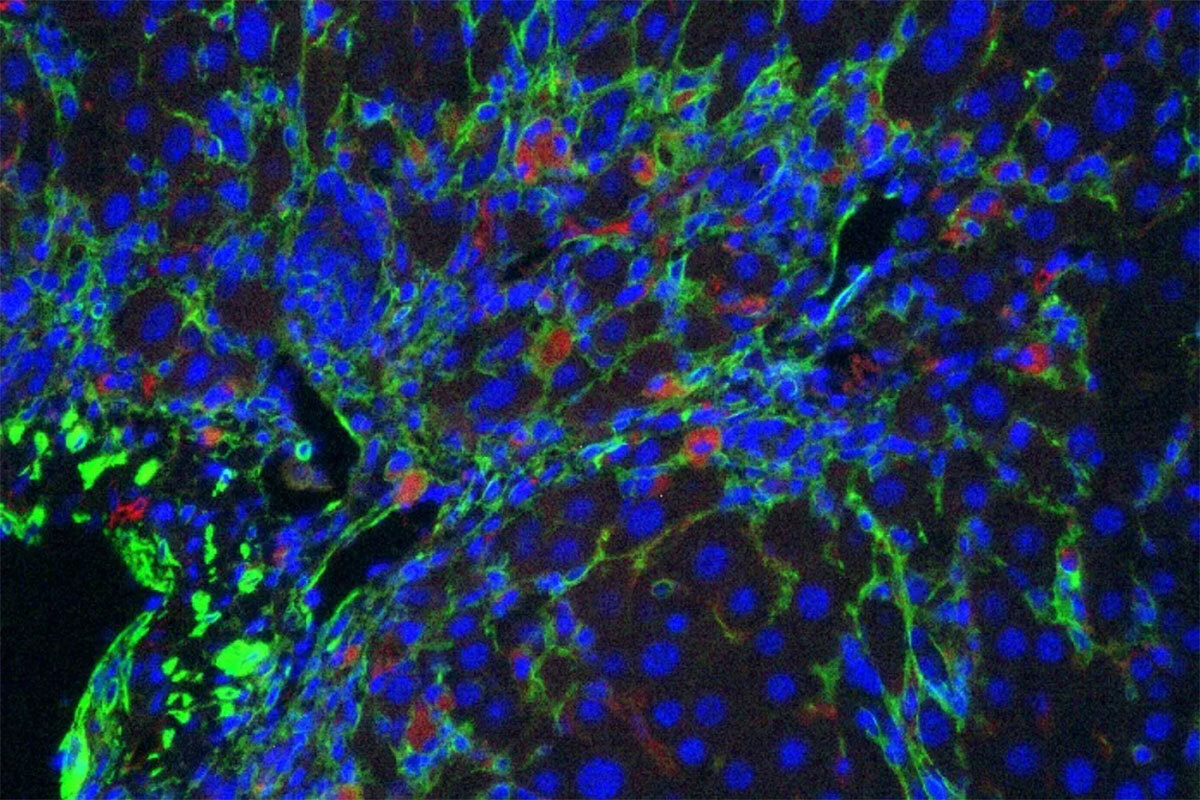
Figure 3. Senolytic CAR T cells. CAR T cells specific for uPAR (red) target senescent cells (green) in a mouse model of liver fibrosis.
References
1. Chicas A, Wang X, Zhang C, McCurrach M, Zhao Z, Mert O, Dickins RA, Narita M, Zhang M, Lowe SW. Dissecting the unique role of the retinoblastoma tumor suppressor during cellular senescence. Cancer Cell. 2010;17(4):376-87.
2. Chien Y, Scuoppo C, Wang X, Fang X, Balgley B, Bolden JE, Premsrirut P, Luo W, Chicas A, Lee CS, Kogan SC, Lowe SW. Control of the senescence-associated secretory phenotype by NF-κB promotes senescence and enhances chemosensitivity. Genes Dev. 2011;25(20):2125-36.
3. Narita M, Narita M, Krizhanovsky V, Nunez S, Chicas A, Hearn SA, Myers MP, Lowe SW. A novel role for high-mobility group a proteins in cellular senescence and heterochromatin formation. Cell. 2006;126(3):503-14.
4. Narita M, Nunez S, Heard E, Narita M, Lin AW, Hearn SA, Spector DL, Hannon GJ, Lowe SW. Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell. 2003;113(6):703-16.
5. Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V, Cordon-Cardo C, Lowe SW. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature. 2007;445(7128):656-60.
6. Kang TW, Yevsa T, Woller N, Hoenicke L, Wuestefeld T, Dauch D, Hohmeyer A, Gereke M, Rudalska R, Potapova A, Iken M, Vucur M, Weiss S, Heikenwalder M, Khan S, Gil J, Bruder D, Manns M, Schirmacher P, Tacke F, Ott M, Luedde T, Longerich T, Kubicka S, Zender L. Senescence surveillance of pre-malignant hepatocytes limits liver cancer development. Nature. 2011;479(7374):547-51.
7. Hoare M, Ito Y, Kang TW, Weekes MP, Matheson NJ, Patten DA, Shetty S, Parry AJ, Menon S, Salama R, Antrobus R, Tomimatsu K, Howat W, Lehner PJ, Zender L, Narita M. NOTCH1 mediates a switch between two distinct secretomes during senescence. Nat Cell Biol. 2016;18(9):979-92.
8. Demaria M, Ohtani N, Youssef SA, Rodier F, Toussaint W, Mitchell JR, Laberge RM, Vijg J, Van Steeg H, Dollé ME, Hoeijmakers JH, de Bruin A, Hara E, Campisi J. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell. 2014;31(6):722-33.
9. Sagiv A, Biran A, Yon M, Simon J, Lowe SW, Krizhanovsky V. Granule exocytosis mediates immune surveillance of senescent cells. Oncogene. 2013;32(15):1971-7.
10. Krizhanovsky V, Yon M, Dickins RA, Hearn S, Simon J, Miething C, Yee H, Zender L, Lowe SW. Senescence of activated stellate cells limits liver fibrosis. Cell. 2008;134(4):657-67.
11. Krtolica A, Parrinello S, Lockett S, Desprez PY, Campisi J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc Natl Acad Sci U S A. 2001;98(21):12072-7.
12. Demaria M, O’Leary MN, Chang J, Shao L, Liu S, Alimirah F, Koenig K, Le C, Mitin N, Deal AM, Alston S, Academia EC, Kilmarx S, Valdovinos A, Wang B, de Bruin A, Kennedy BK, Melov S, Zhou D, Sharpless NE, Muss H, Campisi J. Cellular Senescence Promotes Adverse Effects of Chemotherapy and Cancer Relapse. Cancer Discov. 2017;7(2):165-76.
13. Nicolas AM, Pesic M, Engel E, Ziegler PK, Diefenhardt M, Kennel KB, Buettner F, Conche C, Petrocelli V, Elwakeel E, Weigert A, Zinoveva A, Fleischmann M, Haupl B, Karakutuk C, Bohnenberger H, Mosa MH, Kaderali L, Gaedcke J, Ghadimi M, Rodel F, Arkan MC, Oellerich T, Rodel C, Fokas E, Greten FR. Inflammatory fibroblasts mediate resistance to neoadjuvant therapy in rectal cancer. Cancer Cell. 2022;40(2):168-84 e13.
14. Calls A, Torres-Espin A, Navarro X, Yuste VJ, Udina E, Bruna J. Cisplatin-induced peripheral neuropathy is associated with neuronal senescence-like response. Neuro Oncol. 2021;23(1):88-99.
15. Peng X, Wu Y, Brouwer U, van Vliet T, Wang B, Demaria M, Barazzuol L, Coppes RP. Cellular senescence contributes to radiation-induced hyposalivation by affecting the stem/progenitor cell niche. Cell Death Dis. 2020;11(10):854.
16. Mitry MA, Laurent D, Keith BL, Sira E, Eisenberg CA, Eisenberg LM, Joshi S, Gupte S, Edwards JG. Accelerated cardiomyocyte senescence contributes to late-onset doxorubicin-induced cardiotoxicity. Am J Physiol Cell Physiol. 2020;318(2):C380-C91.
17. Soysouvanh F, Benadjaoud MA, Dos Santos M, Mondini M, Lavigne J, Bertho A, Buard V, Tarlet G, Adnot S, Deutsch E, Guipaud O, Paget V, Francois A, Milliat F. Stereotactic Lung Irradiation in Mice Promotes Long-Term Senescence and Lung Injury. Int J Radiat Oncol Biol Phys. 2020;106(5):1017-27.
18. Childs BG, Durik M, Baker DJ, van Deursen JM. Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med. 2015;21(12):1424-35.
19. Ruscetti M, Leibold J, Bott MJ, Fennell M, Kulick A, Salgado NR, Chen CC, Ho YJ, Sanchez-Rivera FJ, Feucht J, Baslan T, Tian S, Chen HA, Romesser PB, Poirier JT, Rudin CM, de Stanchina E, Manchado E, Sherr CJ, Lowe SW. NK cell-mediated cytotoxicity contributes to tumor control by a cytostatic drug combination. Science. 2018;362(6421):1416-22.
20. Ruscetti M, Morris JP 4th, Mezzadra R, Russell J, Leibold J, Romesser PB, Simon J, Kulick A, Ho YJ, Fennell M, Li J, Norgard RJ, Wilkinson JE, Alonso-Curbelo D, Sridharan R, Heller DA, de Stanchina E, Stanger BZ, Sherr CJ, Lowe SW. Senescence-induced vascular remodeling creates therapeutic vulnerabilities in pancreas cancer. Cell. 2020;181(2):424-41 e21.
21. Chen HA, Ho YJ, Mezzadra R, Adrover JM, Smolkin R, Zhu C, Woess K, Bernstein N, Schmitt G, Fong L, Luan W, Wuest A, Tian S, Li X, Broderick C, Hendrickson RC, Egeblad M, Chen Z, Alonso-Curbelo D, Lowe SW. Senescence rewires mcroenvironment sensing to facilitate antitumor immunity. Cancer Discov. 2023;13(2):432-53.
22. Ruscetti M, Morris JP, Mezzadra R, Russell J, Leibold J, Romesser PB, Simon J, Kulick A, Ho YJ, Fennell M, Li J, Norgard RJ, Wilkinson JE, Alonso-Curbelo D, Sridharan R, Heller DA, de Stanchina E, Stanger BZ, Sherr CJ, Lowe SW. Senescence-Induced Vascular Remodeling Creates Therapeutic Vulnerabilities in Pancreas Cancer. Cell. 2020;181(2):424-41 e21.
23. Basisty N, Kale A, Jeon OH, Kuehnemann C, Payne T, Rao C, Holtz A, Shah S, Sharma V, Ferrucci L, Campisi J, Schilling B. A proteomic atlas of senescence-associated secretomes for aging biomarker development. PLoS Biol. 2020;18(1):e3000599.
24. Wiley CD, Velarde MC, Lecot P, Liu S, Sarnoski EA, Freund A, Shirakawa K, Lim HW, Davis SS, Ramanathan A, Gerencser AA, Verdin E, Campisi J. Mitochondrial dysfunction induces senescence with a distinct secretory phenotype. Cell Metab. 2016;23(2):303-14.
25. Chibaya L, Murphy KC, DeMarco KD, Gopalan S, Liu H, Parikh CN, Lopez-Diaz Y, Faulkner M, Li J, Morris JP 4th, Ho YJ, Chana SK, Simon J, Luan W, Kulick A, de Stanchina E, Simin K, Zhu LJ, Fazzio TG, Lowe SW, Ruscetti M. EZH2 inhibition remodels the inflammatory senescence-associated secretory phenotype to potentiate pancreatic cancer immune surveillance. Nat Cancer. 2023;4(6):872-92.
26. Paffenholz SV, Salvagno C, Ho YJ, Limjoco M, Baslan T, Tian S, Kulick A, de Stanchina E, Wilkinson JE, Barriga FM, Zamarin D, Cubillos-Ruiz JR, Leibold J, Lowe SW. Senescence induction dictates response to chemo- and immunotherapy in preclinical models of ovarian cancer. Proc Natl Acad Sci U S A. 2022;119(5).
27. Amor C, Feucht J, Leibold J, Ho YJ, Zhu C, Alonso-Curbelo D, Mansilla-Soto J, Boyer JA, Li X, Giavridis T, Kulick A, Houlihan S, Peerschke E, Friedman SL, Ponomarev V, Piersigilli A, Sadelain M, Lowe SW. Senolytic CAR T cells reverse senescence-associated pathologies. Nature. 2020;583(7814):127-32.
28. Amor C, Fernandez-Maestre I, Chowdhury S, Ho YJ, Nadella S, Graham C, Carrasco SE, Nnuji-John E, Feucht J, Hinterleitner C, Barthet VJA, Boyer JA, Mezzadra R, Wereski MG, Tuveson DA, Levine RL, Jones LW, Sadelain M, Lowe SW. Prophylactic and long-lasting efficacy of senolytic CAR T cells against age-related metabolic dysfunction. Nat Aging. 2024;4(3):336-49.