Greasing the gears: Discoveries in basic science are crucial for making progress in cancer treatment and prevention. These often incremental breakthroughs are pivotal for driving clinical advances forward.
This past year, Memorial Sloan Kettering researchers made important advances in basic and translational science that furthered our understanding of cancer and of how best to treat it. Here, we describe some of the year’s highlights.
First CAR T Cell Therapies FDA Approved
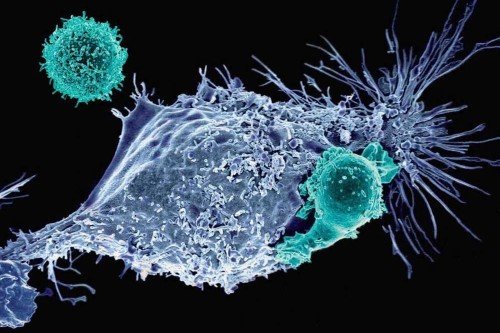
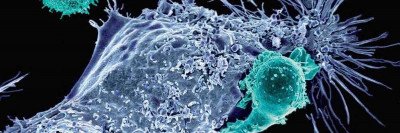
By far the biggest cancer-science news from 2017 was the US Food and Drug Administration’s approval of chimeric antigen receptor (CAR) T cell therapy for certain types of leukemia and lymphoma. A form of immunotherapy, CAR T uses genetically engineered versions of a person’s own immune cells to find and fight cancer.
The FDA called its approval “historic,” noting that it was the first time that a cell therapy produced through gene editing had passed this regulatory hurdle.
Two separate CAR T products were given the green light within two months. In August, the FDA approved tisagenlecleucel (Kymriah), made by the pharmaceutical company Novartis, for children and young adults with acute lymphoblastic leukemia that has not been completely eliminated by prior therapies. In October, it approved axicabtagene ciloleucel (Yescarta) for people with certain types of non-Hodgkin lymphoma that are similarly resistant to treatment. Yescarta is manufactured by the biotech company KITE, now owned by Gilead Sciences. Both approvals reflect the significant benefits of CAR T therapies for very ill people who otherwise have run out of options.
MSK scientists played key roles in the development of CAR T cell therapy, and they are leaders in pioneering the next generation of these medicines. Read more about MSK’s contributions to CAR T therapy.
CRISPR Takes Off
No biotechnology has had more impact on science and medicine in recent years than the genome-editing tool CRISPR. With this powerful molecular scissors, scientists can snip out specific pieces of DNA or make changes at precise genetic addresses. MSK scientists used CRISPR in several inventive ways this year to push cancer science forward.
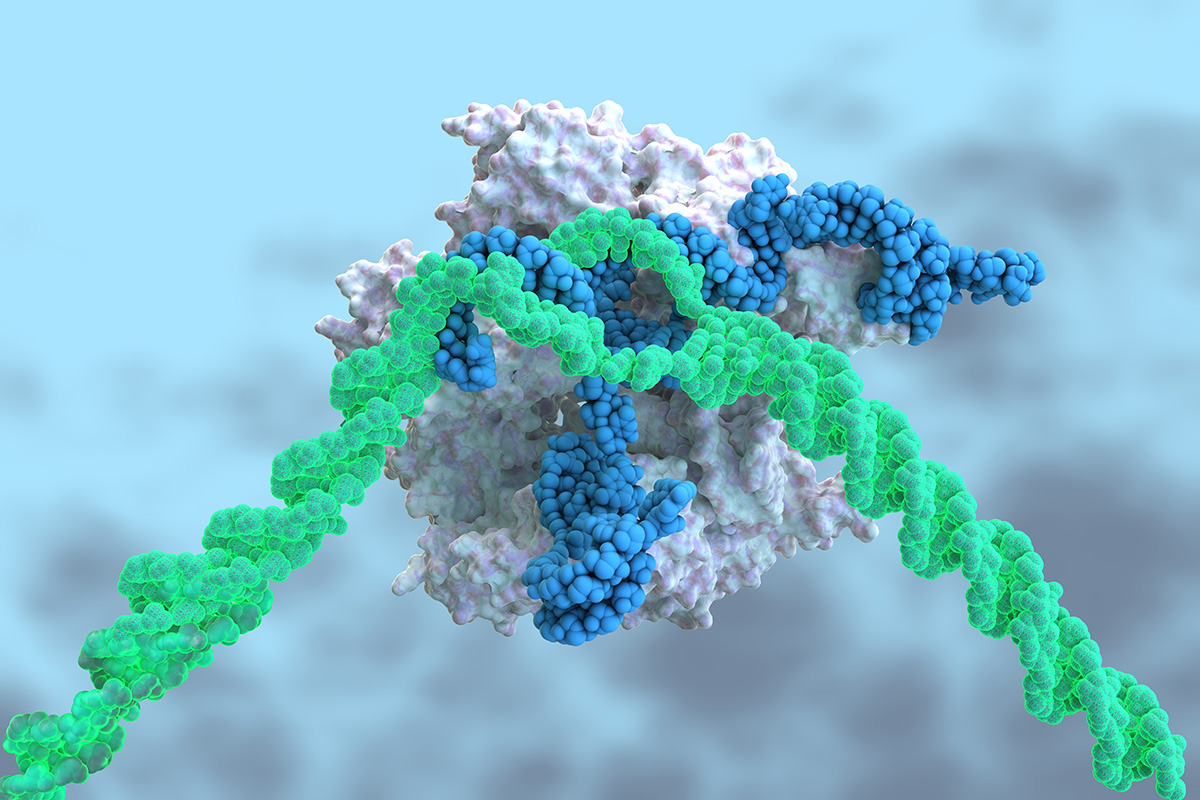
Scientists at the Sloan Kettering Institute have created software to aid researchers using the popular genome-editing tool CRISPR, represented above in white and blue, separating strands of DNA, shown in green.
Immunologist Michel Sadelain and his team used CRISPR to build more-effective CAR T cells. Typically, scientists use modified viruses to add the CAR gene into immune cells. The virus inserts the gene randomly at multiple spots in the genome, which can have unwanted consequences. In a study published in the journal Nature, Dr. Sadelain’s team showed that they could use CRISPR to place the CAR at a very specific genome location, to optimize functioning. The approach paves the way for more reliable CAR therapies, and even off-the-shelf versions that would not need to be made from a person’s own cells.
In a separate study, biologist Scott Lowe and his team used CRISPR to prove that a specific genetic mutation is the cause of a rare type of liver cancer called fibrolamellar hepatocellular carcinoma. The discovery should aid the search for new medicines that could treat this rare cancer, which predominantly affects children and young adults.
As if CRISPR weren’t precise enough, a team of researchers from the Sloan Kettering Institute built a computer program to help scientists design more-effective guide RNAs, which is what the CRISPR tool uses to find specific regions of the genome. Called GuideScan, the freely accessible program should help researchers make even more skillful use of the technology.
This past year, MSK researchers also used CRISPR to study how acute myeloid leukemia progresses and to create models of pancreatic cancer.
Jumping Ahead of Childhood Cancer
Cancer primarily affects older people. A person’s risk rises over time as genetic mutations pile up, due either to mistakes when cells copy their DNA or to ongoing exposure to cancer-causing agents in the environment, like cigarettes. Researchers have long been puzzled by why tumors develop in children, who presumably haven’t had enough time for large numbers of random mutations to accumulate.
This year, a team of MSK scientists led by Alex Kentsis discovered a surprising cause of childhood cancers: a gene called PGBD5 that becomes abnormally activated in children. The protein made by the gene snips out DNA segments and flips them or moves them to a different location within the genome. Scientists have dubbed these mobile elements “jumping genes.” When they jump, these DNA bits can land in other important genes, producing mutations that lead to cancer.
In a related study, also published this year, Dr. Kentsis’s team turned their discovery into a potential new treatment strategy. When the PGBD5 protein scrambles other genes, it creates breaks in the DNA that must continuously be fixed in order for the cell to survive. This damage causes cells with a lot of PGBD5 to be especially dependent on the processes of DNA repair. Therefore, blocking DNA repair in these cells with drugs could be an effective way to treat these cancers.
Plugging a Hole in Metastasis
Around 90% of cancer deaths are caused by metastasis — when cancer spreads from one part of the body to another. One of the most challenging types of metastatic cancer is leptomeningeal metastasis (LM). It occurs when cancer cells spread to the fluid and tissues that surround the brain and spine. Once LM arises, most patients die within a few weeks or months.
How cancer cells can survive and grow in spinal fluid, where there are no nutrients to support their growth, has long been a mystery. But this year, a team from MSK solved it. Physician-scientist Adrienne Boire, working in collaboration with Sloan Kettering Institute Director Joan Massagué, discovered that LM cancer cells make a protein that essentially punches holes in the membrane separating the spinal fluid from the blood supply. These holes then allow growth factors and other nutrients from the blood to enter.
The protein is well known to researchers who study inflammation. Called C3, it is part of a system of blood proteins. Medications developed to block this protein may help to plug the leak in the membrane and could be a way to treat this deadly cancer complication.
Understanding “Exhausted” Immune Cells
Like any overworked laborer, cancer-fighting immune cells eventually become exhausted. When this occurs, they lose their ability to effectively combat the disease. If there were a way to revive these sluggish cells, immune-based cancer therapies might be more effective.
Immunologist Andrea Schietinger and her team studied the biology of these exhausted T cells and discovered characteristic changes in the way that their DNA is packaged. These changes — which are referred to as epigenetic because they don’t involve changes to the DNA sequence itself — reveal new targets for drugs aimed at making immunotherapies more effective for more people.
Immunotherapy drugs called checkpoint inhibitors have revolutionized the treatment of certain cancers. But despite their promise, these drugs are effective in only a fraction of patients. The epigenetic states Dr. Schietinger’s team has identified might help predict which patients will respond to immunotherapy, and what else can be done to increase the number of people who do respond.
Dr. Schietinger received a prestigious New Innovator Award from the National Institutes of Health for her research. The award supports exceptionally creative early career investigators who propose innovative, high-impact projects.