Parkinson’s disease
Our lab has worked for more than a decade on developing a cell therapy for Parkinson’s disease (PD). We have tested many types of stem cells over the years as a potential source of dopamine neurons. However, only recently have we resolved some of the major challenges in generating the correct nerve cell type at sufficient scale and purity. Using this novel differentiation strategy we have demonstrated that lab-grown dopamine nerve cells can efficiently engraft and function in mouse, rat or monkey models of PD.
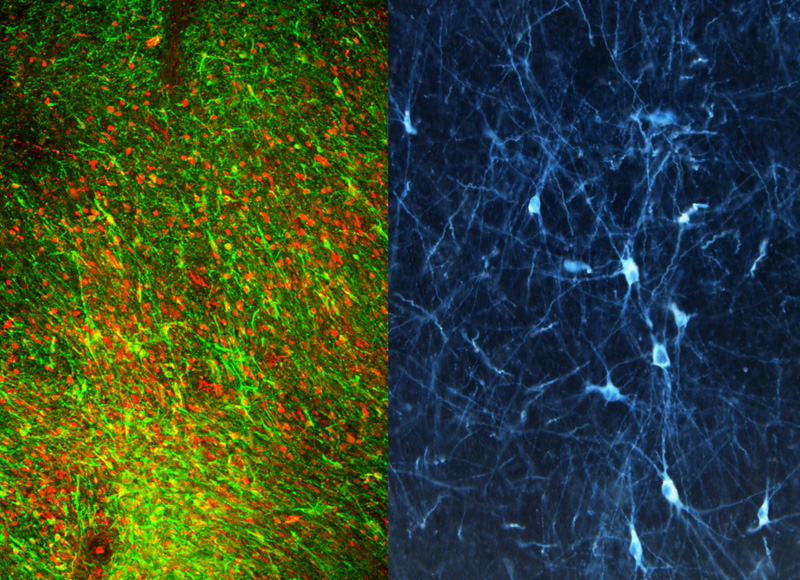
In addition to pursuing the first clinical application of human ESC-derived dopamine neurons, we continue to work on detailing the mechanisms of graft function by implementing novel tools such as optogenetics, a technology that enables “switching off” or “switching on” grafted cells in freely moving animals. Using genetic reporter tools and surface marker screening technology, we are also further defining the optimal composition of a midbrain dopamine neuron graft. Insights from those studies should go toward further improving cell therapy for PD in the future.
Other CNS & PNS disorders
We are interested in applying our ability to generate disease-relevant cell types and in establishing clinical-grade protocols for the development of cell therapy approaches in other CNS and PNS disorders. Collaborative studies are ongoing to explore the potential of hPSC derivatives such as oligodendrocytes, Schwann cell precursors, and peripheral neurons.