Stem cell therapy for Parkinson’s disease has shown great promise in animal studies and is moving closer to becoming an option for people affected by this devastating neurological condition. But before it can advance from animal models into human trials, investigators need to learn more about how the therapy works.
Memorial Sloan Kettering stem cell biologist Lorenz Studer and his colleagues have taken a critical step in that direction with their latest study, which provides an unprecedented glimpse into the brains of mice treated with engineered stem cells. Using optogenetics — a tool that controls cells by shining light on them — they’ve been able to decipher how these transplanted cells function in the brain.
“Optogenetics has had a huge impact in neurobiology over the past several years by allowing researchers to study how parts of the brain interact with each other,” Dr. Studer says. “But to my knowledge no one else has used this technology to find out how newly grafted cells in the brain function. Now we have a way to understand how the cells really work.”
Manufacturing Stem Cells
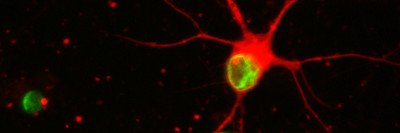
For more than a decade, Dr. Studer’s lab has focused on developing embryonic stem cells as a treatment for Parkinson’s disease, a degenerative disorder caused by the loss of neurons in the brain that produce the hormone dopamine. Dr. Studer and his colleagues have created methods for coaxing embryonic stem cells — which have the potential to form any type of cell in the body — into becoming dopamine neurons by guiding their development.
Using laboratory mice with a Parkinson’s-like disease, the investigators have shown that transplanting these neurons into the brains of the mice alleviates the movement-related symptoms — such as shaking and difficulty moving — that characterize the illness.
“When we put these cells into the brains of a Parkinson’s animal, it takes about four or five months, but eventually the animal recovers from its movement deficits,” Dr. Studer explains. “This is exciting, but the question has been, how do the cells do it? Do they induce the remaining dopamine cells in the brain to function better? Do they perform some kind of signaling function? Or do they actually integrate into the network of the host animal and begin acting like a new part of the brain? This technique allows us to answer those questions.”
Using Optogenetics to Look Inside the Brain
In the current study, which was published earlier this month in Nature Biotechnology, the team introduced a genetic switch into the stem cell–derived dopamine neurons that would allow the scientists to turn the firing of nerve cells on or off at will by exposing them to light. This manipulation is the basis of optogenetics.
The team, including researchers from Columbia University and Stanford University, was able to show that once the mice had fully recovered from their motor disease symptoms after transplantation, their Parkinson’s-like ailments would return again within minutes if the scientists switched on a laser light that inactivated the newly transplanted cells. When the light was switched off, the animals fully recovered once again.
“This is quite a remarkable demonstration that the grafted cells need to constantly be active through neuronal firing to provide the therapeutic benefit to the animals,” Dr. Studer says. “We also showed that this neuronal activity is linked to increased dopamine release from the grafted cells, indicating that these implanted cells truly integrate into the animal’s brain.”
Advancing to Human Trials
“Our thinking in developing this research was that if you’re going to treat patients with these neurons, you should first be able to prove exactly how these nerve grafts work,” says Julius Steinbeck, a postdoctoral researcher in Dr. Studer’s lab who was the study’s first author.
“Understanding how this treatment works will also help us to anticipate potential side effects before we move this into human studies,” he adds.
“There’s still more to learn from studying these implanted cells,” Dr. Studer concludes. “We plan to continue using this tool to get more details about how the grafted cells function.”
This work was supported by the US National Institutes of Health and its National Institute of Neurological Disorders and Stroke, a New York State Stem Cell Science contract, the Parkinson’s Disease Foundation, the JPB Foundation, and the Morris K. Udall Center of Excellence for Parkinson’s Disease Research.