Molecular Pharmacology Program
The David Scheinberg Lab
Research

Professor
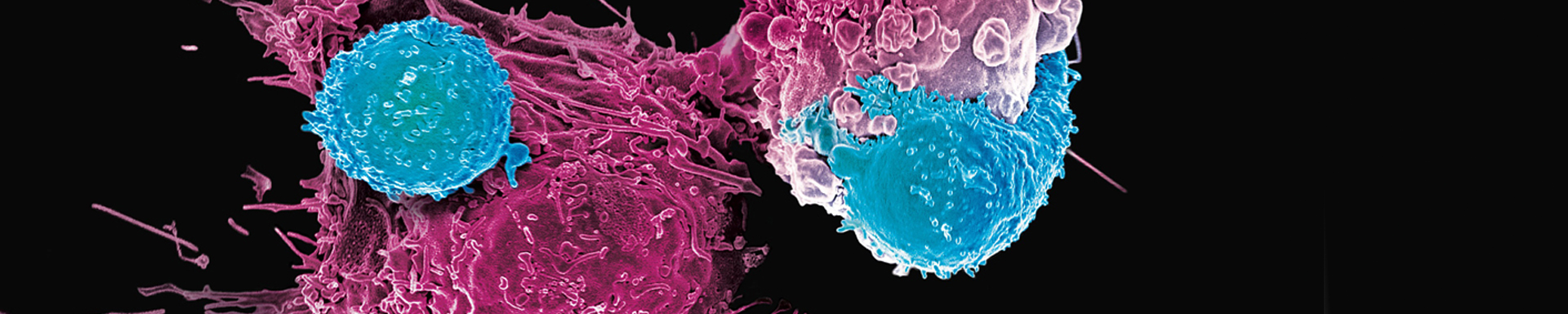
The overall goals of this laboratory are to develop novel targeted immunotherapies based on effectors of the immune system and to understand their mechanisms of action as well as the mechanisms of resistance to them. This includes antibodies, targeted nano-devices, engineered cells, and active specific agents such vaccines. An important goal is to take these new therapies into human clinical trials for testing. Examples include a molecular nanogenerator that releases alpha particles inside cancer cells, now being tested in Phase II human trials (Blood, 2010), and a vaccine to WT1 in Phase III trials. Read full article in Science.
We have developed targeted alpha particle therapies, targeted beta emitters, oncogenic protein peptide vaccines, humanized antibodies, each of which reached human clinical trials; More recently we have discovered TCR mimic antibodies to intracellular proteins (Science Trans. Med., 2013), (Nature Biotech., 2015), as well as prototype targeted nano-machines (Nature Nano, 2013), and cellular micropharmacies (Nature Chem.Biol., 2021).
Research Projects

Featured News


Publications Highlights
Dao T, Pankov D, Scott A, Korontsvit T, Zakhaleva V, Xu Y, Xiang J, Yan S, de Morais Guerreiro MD, Veomett N, Dubrovsky L, Curcio M, Doubrovina E, Ponomarev V, Liu C, O’Reilly RJ, Scheinberg DA. Therapeutic bispecific T-cell engager antibody targeting the intracellular oncoprotein WT1. Nature biotechnology. 2015;33(10):1079-86. Doi: 10.1038/nbt.3349. PubMed PMID: 26389576; PMCID: PMC4600043.
People

David A. Scheinberg, MD, PhD
Chair, Center for Experimental Therapeutics, MSK; Deputy Director, Sloan Kettering Institute, for Therapeutic Discovery
Professor
- Molecular Pharmacology Program Member David Scheinberg focuses on the discovery and development of novel, specific immunotherapeutic agents and targeted nanodevices for cancer therapy.
- MD, PhD, Johns Hopkins University School of Medicine
- [email protected]
- Email Address
- 646-888-2190
- Office Phone
- View physician profile
- Physician profile
Members


- PhD: Immunology, Hyogo College of Medicine, Japan
- MD: Medicine, Medical School of Inner Mongolia
- Postdoctoral: Immunology, Yale Medical School Immunobiology.


- PhD in Chemistry from Case Western Reserve University in Cleveland, Ohio (1985)
- ME in Chemical Engineering from The City University of New York (2004)

- BS in Biophysics from Humboldt University in Berlin (2018)
- MS in Biomedical Sciences from UCL London (2019)

- B.A. in Biochemistry from Wellesley College, MA

- BS in Biochemistry/Molecular Biology (Friedrich Schiller University Jena)
- MS in Biochemistry (University of Tübingen)
- PhD in Biology (Memorial Sloan Kettering Cancer Center/University of Freiburg)
- 646-888-2205
- Office Phone

- University of California, Berkeley (2021)


- University of Michigan

- BS in Biochemistry and Molecular Biology from The University of Georgia, Athens, GA, 2018

- University of Texas at Austin (Undergraduate)
- Weill Cornell Medicine (Currently enrolled, class of 2025, MD in progress)

- MD in Internal Medicine from Sechenov (1st) Moscow Medical Institute, Moscow, Russia,(1990)
- PhD in Medical Science, from State Scientific Center of Russian Federation Institute of Biophysics, Moscow, Russia,(1997)

- BS

- MSc in Molecular Biotechnology from Ruprecht-Karls University of Heidelberg, Germany, 2021
- BSc in Molecular Biotechnology from Ruprecht-Karls University of Heidelberg, Germany, 2017


















- BS, Cornell University




- BA in Biology from Swarthmore College, Swarthmore, PA (2017)



- BS in Biomedical Sciences at Utrecht University
- MS in Biomedical Sciences/Biology of Disease at Utrecht University
- PhD in Pediatric Cancer Immunology at Utrecht University
- Postdoc in Pediatric Cancer Cellular Therapy and Immunology at Princess Máxima Center for Pediatric Oncology

- BS in Biomedical Engineering from Boston University (2016)

- PhD in Oncology from A. C. Camargo Hospital, Sao Paulo, BR
- and from MD Anderson Cancer Center, Texas US (2009)


- MD from Eberhard Karls University in Tuebingen, Germany (2014)

- MD from University of Central Florida College of Medicine, 2016

- PhD in Immunology from Heinrich Heine University, Germany (2015)

- BS in Biology from St. John' s University in Queens, NY (2017)

- PhD in Chemistry from Tufts University (2018)
- BS in Biochemistry from Hobart and William Smith Colleges (2013)

- BS in Chemistry from The College of New Jersey, Ewing NJ (2019)






























- Molecular Pharmacology Program
- Department of Medicine at MSK
- Center for Experimental Therapeutics
- Center for Cell Engineering
- Geoffrey Beene Cancer Research Center
- Center for Molecular Imaging & Bioengineering
- Tri-Institutional PhD Program in Chemical Biology
- Gerstner Sloan Kettering Graduate School of Biomedical Sciences
Lab News & Events
Open Positions
To learn more about available postdoctoral opportunities, please visit our Career Center
To learn more about compensation and benefits for postdoctoral researchers at MSK, please visit Resources for Postdocs
Get in Touch
-
Lab Head Email
-
Office Phone
-
Lab Phone
To learn more about Postdoc compensation and benefits at MSK, please visit https://www.mskcc.org/education-training/postdoctoral/resources-postdocs/compensation-benefits-resources
Disclosures
Members of the MSK Community often work with pharmaceutical, device, biotechnology, and life sciences companies, and other organizations outside of MSK, to find safe and effective cancer treatments, to improve patient care, and to educate the health care community. These activities outside of MSK further our mission, provide productive collaborations, and promote the practical application of scientific discoveries.
MSK requires doctors, faculty members, and leaders to report (“disclose”) the relationships and financial interests they have with external entities. As a commitment to transparency with our community, we make that information available to the public. Not all disclosed interests and relationships present conflicts of interest. MSK reviews all disclosed interests and relationships to assess whether a conflict of interest exists and whether formal COI management is needed.
David A. Scheinberg discloses the following relationships and financial interests:
-
Actinium
Equity; Intellectual Property Rights; Professional Services and Activities (Uncompensated) -
Arvinas, Inc.
Equity -
Atengen, Inc.
Equity; Professional Services and Activities (Uncompensated) -
Century Therapeutics
Intellectual Property Rights -
Epics Therapeutics S.A.
Professional Services and Activities -
Estrella Biopharma, Inc.
Professional Services and Activities -
Eureka Therapeutics Inc
Equity; Professional Services and Activities (Uncompensated) -
Great Point Partners
Equity; Professional Services and Activities
-
Iovance Biotherapeutics, Inc.
Equity -
Lantheus Medical Imaging, Inc.
Equity -
Lilly Oncology
Equity -
Omnipotent Bio Holdings, Inc.
Equity; Professional Services and Activities -
Pfizer, Inc.
Equity -
Repertoire Immune Medicines, Inc.
Professional Services and Activities -
Sapience Therapeutics, Inc.
Equity; Fiduciary Role / Position -
Sellas Life Science Group
Equity; Fiduciary Role / Position; Intellectual Property Rights
The information published here is a complement to other publicly reported data and is for a specific annual disclosure period. There may be differences between information on this and other public sites as a result of different reporting periods and/or the various ways relationships and financial interests are categorized by organizations that publish such data.
This page and data include information for a specific MSK annual disclosure period (January 1, 2024 through disclosure submission in spring 2025). This data reflects interests that may or may not still exist. This data is updated annually.
Learn more about MSK’s COI policies here. For questions regarding MSK’s COI-related policies and procedures, email MSK’s Compliance Office at [email protected].

