Developmental biologist Mary Baylies studies the mechanisms that form and maintain muscle both during normal development and in disease.
Molecular Mechanisms of Development
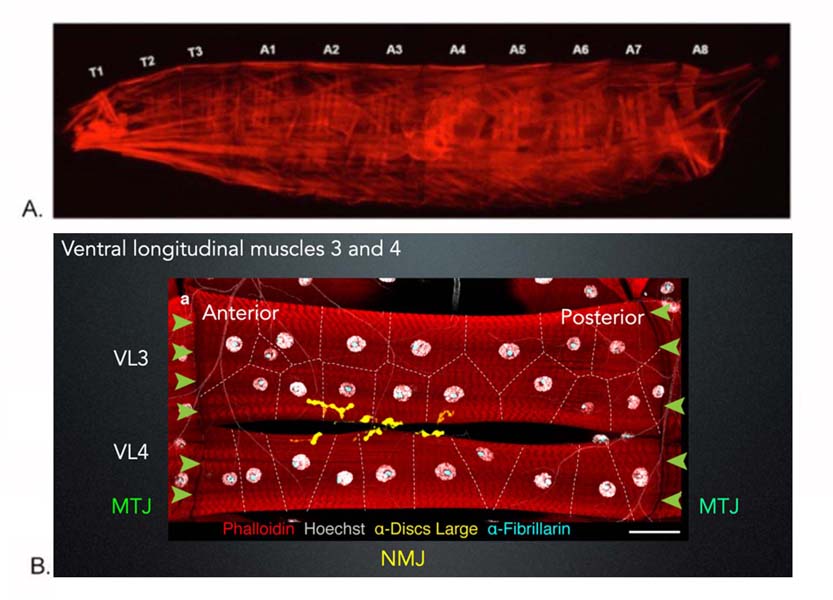
Skeletal muscles come in a variety of shapes and sizes, each tuned to a particular function — from the muscles that allow a person to run to those that focus the eye. Individual muscles also have different susceptibilities to diseases like muscular dystrophy, muscle myopathies, and rhabdomyosarcoma.
Therapies that repair muscle wasting due to aging or disease require the ability to generate specific muscle types of a particular size and shape. Therefore, identifying the mechanisms of how muscles differentiate, as well as how they grow and respond to disease, is critical. My lab focuses on the following questions:
- How are muscle cells specified?
- How are they maintained during growth and use?
- How do they respond and change during disease?
Larval Crawling Assay
Our primary model system is the Drosophila musculature, in which we employ genetics, molecular and cell biological approaches, bioinformatics, in vivo time-lapse imaging, confocal and super resolution microscopy, mathematical modeling, single nuclear sequencing, and biochemistry to identify and characterize genes required for muscle formation, use, and maintenance. (See Figures 1, 2). We currently are generating human skeletal muscle from human pluripotent stem cells. We will use this model to confirm and extend the paradigms that we develop in Drosophila.
Currently, we study two significant areas of muscle biology:
- Fundamental mechanisms governing muscle formation and maintenance. These projects include investigation of muscle identity, muscle cell size, and muscle subcellular organization and myonuclear identity.
- Mechanisms underlying muscle wasting. These projects investigate disease progression and novel therapeutics for conditions in which muscle is comprised, including diseases such as rhabdomyosarcoma, cancer cachexia, and nemaline myopathy, as well as in aging.