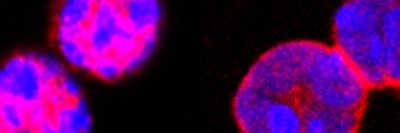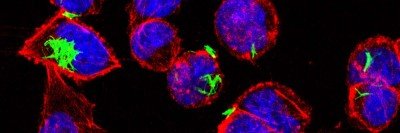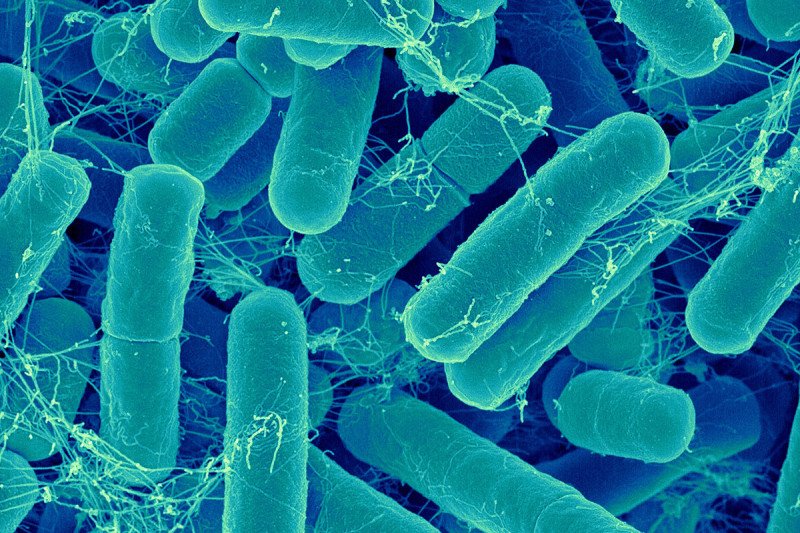
The microscopic organisms that live in our intestines perform many jobs to keep us healthy, such as helping our bodies digest nutrients from food. They keep harmful microbes under control and prevent them from taking over the gut, and also play a role in regulating the immune system.
Researchers at Memorial Sloan Kettering are studying the connections between gut microbes and human health. In a study published April 15 in Nature, an MSK team uncovered new findings about an important relationship: the way that gut microbes promote the formation of a type of immune cell called regulatory T cells (Tregs).
Tregs help keep powerful immune responses in check. They are crucial for preventing autoimmunity, when the immune system mistakenly attacks the body. Tregs also play a role in cancer: Cancer is more likely to develop when malfunctioning Tregs lead to chronically inflamed tissue. This is seen in conditions such as inflammatory bowel disease (IBD).
Focusing on the Microbe–T Cell Connection
“Life in all mammals, including humans, is impossible without regulatory T cells,” says the study’s senior author, Alexander Rudensky, Chair of the Immunology Program in the Sloan Kettering Institute and a Howard Hughes Medical Institute investigator. “This study builds on previous research from our lab and others that looked at how these cells are made and why they are so important.
“It’s already been shown that disturbance of the microbial community in the gut is associated with autoimmune and inflammatory diseases, like IBD,” he adds. “These findings have illustrated the importance of the relationship between the microbes that live in our guts and our immune systems.”
To better understand this relationship, Dr. Rudensky and his team study how different microbes in the gut affect the production of Tregs that protect against inflammation and autoimmune conditions. They do this by looking at the molecules that microbes make to carry out their metabolic functions. Then they study how those metabolites in turn influence the manufacturing of Tregs.
In the experiments, the investigators concentrated on a class of metabolites called secondary bile acids. These are produced by gut microbes. Bile acids help the digestion of dietary fats.
Members of Dr. Rudensky’s team focused on a type of gut bacteria belonging to a group called Bacteroides and engineered them to produce a particular bile acid, called isoDCA. They exposed naive T cells, which had not yet developed into a specific T cell type, to isoDCA during the process of their activation. They found that isoDCA caused the immature T cells to become Tregs.
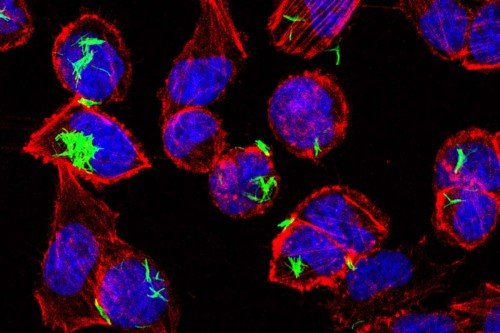
Ultimately, these findings could lead to a bacterial-based therapy for the treatment of IBD associated with imbalanced microbes and diminished Treg activity. (Currently, IBD is treated with anti-inflammatory drugs, which can have many side effects.) But Dr. Rudensky says much more work is needed before this kind of treatment could be tested. Many parts of the process are still not well understood. In addition, researchers would have to do extensive testing in mice before designing a clinical trial. “We plan to continue exploring the possibility of using metabolic products from microbes for the treatment of inflammatory disorders,” he says.
The two first authors on the paper were research fellow Clarissa Campbell and research associate Peter McKenney in Dr. Rudensky’s lab. Investigators in MSK’s Donald B. and Catherine C. Marron Cancer Metabolism Center, including the center’s director, Justin Cross, also contributed to the research.
This study was funded by National Institutes of Health grants (R01 AI034206 and 1DP2HD101401-01), the Ludwig Institute for Cancer Immunotherapy, the Hilton Foundation, Research Beyond Borders at Boehringer Ingelheim, and the Howard Hughes Medical Institute.
Dr. Rudensky is a cofounder of Vedanta Biosciences.