Peyton Rous wasn’t expecting a chicken to show up in his laboratory one day in 1909. But there she was, a barred Plymouth Rock hen with a bright red comb, clucking amidst the Bunsen burners.
The chicken’s owner, a poultry farmer from Long Island, had noticed a lump protruding from the bird’s right breast. Concerned that she might sicken other members of his flock, the farmer turned to Dr. Rous for help.
A pathologist at the Rockefeller Institute in New York City, Dr. Rous was known for having an interest in cancer. He took a biopsy of the lump and found it to be a sarcoma, a type of cancer that affects muscle or bone tissue. What he did next changed the course of cancer treatment.
Dr. Rous cut off a piece of the tumor, ground it up, and passed it through a filter to remove all cells. Then he injected this cell-free liquid into other chickens. Within a few weeks, they too developed tumors.
“That was really the first hint that cancer, or a tumor, could be transmitted by a microscopic, noncellular agent — what we now call a virus,” says Marilyn Resh, a cell biologist at the Sloan Kettering Institute.
The discovery was remarkable, since cancers in humans weren’t known to be contagious. In fact, the result was so surprising that few contemporaries paid much attention to it. Not until many years later did scientists recognize the significance of what Dr. Rous had found. In honor of the breakthrough, he was awarded a Nobel Prize in 1966. The virus he discovered became known as the Rous sarcoma virus (RSV).
But that was really just the beginning of a long line of breakthroughs hatched by this bird tumor. When it comes to cancer research, Dr. Rous’s chicken was the gift that kept on giving.
Funky Chicken Virus
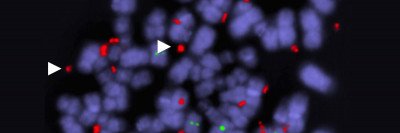
The rediscovery of Dr. Rous’s work in the 1960s led researchers to ask what it was about the virus that enabled it to cause cancer. From early experiments, they knew that every cell in the chicken tumor contained the Rous sarcoma virus. This implied that the virus was necessary to keep cells in a cancerous state. And, since it was present in every cancer cell, this seemed to suggest it had smuggled itself into the cell’s own DNA.
By 1970, scientists had homed in on one piece — one gene — of the viral genome that seemed responsible for the cancer-promoting activity of the virus. They dubbed it src (pronounced “sarc,” short for sarcoma). But they were thrown for a loop when they learned that the virus’s genetic material is made of RNA, not DNA.
“Initially, nobody knew what to do with that because the dogma was that information in cells flows from DNA to RNA to protein,” says Dr. Resh. “It wasn’t clear how you could get from RNA to DNA.”
Biologist Howard Temin from the University of Wisconsin, along with David Baltimore from the Massachusetts Institute of Technology, eventually offered a solution. In 1970, they discovered that RSV contains an enzyme called reverse transcriptase that transcribes RNA into DNA — the exact reverse of what normally happens in cells. This DNA copy of the viral genome is then incorporated into the cell’s own DNA. The two scientists shared the Nobel Prize for this work in 1975. (If you’re keeping score, that’s two Nobel Prizes so far for our chicken.)
RSV was the first retrovirus to be discovered. We now know that there are many retroviruses that affect human health. Perhaps the most famous is HIV.
The Oncogene as Genetic Hitchhiker
The discovery that a viral gene could cause cancer when it inserted itself into cells’ DNA led to the idea that our genomes might be full of cancer genes derived from viruses. Scientists termed them oncogenes. Perhaps these viral genes were genetic holdovers from past infections, and cancer resulted when these viruses “woke up” and wreaked havoc.
That was the idea, anyway. But a series of discoveries made by the team of Harold Varmus and J. Michael Bishop from the University of California, San Francisco, pointed to a different conclusion. Dr. Varmus (who would eventually become MSK’s president) and Dr. Bishop were struck by the fact that a gene resembling src was present in many different birds, not just chickens. They found it in ducks, turkeys, quail — even an emu from the San Francisco Zoo.
To them, this suggested that the src gene was not originally a viral gene at all, but rather a normal cellular gene that RSV had picked up in the course of its travels. In other words, it was a genetic hitchhiker.
The radical implication of this finding was that cancer isn’t only or even primarily a disease caused by outside invaders. The danger was lurking in our own genes all along.
Additional research confirmed that a gene related to src is present in essentially all animals, from flies to worms to humans. For the discovery of the cellular origin of oncogenes, Drs. Bishop and Varmus were awarded the 1989 Nobel Prize in Physiology or Medicine, bringing the total of RSV-related Nobels up to three.
When Bad Genes Come Home to Roost
Why would normal cells carry genes that cause cancer? Scientists eventually learned that src makes a type of protein called a kinase. Kinases are enzymes — proteins that speed up chemical reactions. They remove phosphates (P) from ATP, the cell’s energy currency, and attach them to other proteins, changing their activity. This sticking-on of phosphates is called phosphorylation. Kinases help control growth and reproduction in cells, among other behaviors. Normally, they are active for a time, then the cell switches them off.
The mutant version of the src gene, found in RSV, makes a kinase that is essentially on all the time, like a gas pedal stuck to the floor. “The problem with the onco-proteins is that they just keep phosphorylating and phosphorylating,” Dr. Resh says.
Getting infected with RSV is one way to acquire this mischievous oncogene. Another is for a random mutation to occur in one or both of the cell’s own copies of this gene.
Since src was discovered, scientists have found numerous other oncogenes in cells. Many, like src, encode protein kinases. In their normal form, they are referred to as proto-oncogenes; in their mutated (cancer-causing) form, they are called oncogenes. Common ones include myc, EGFR, Her2/neu, and BCR-ABL.
Knowing which genes and proteins are malfunctioning to cause cancer opens the door to targeted therapies directed at these bad actors. The first such targeted therapy was imatinib (Gleevec®), which blocks the action of the BCR-ABL onco-protein.
“There’s an enormously rich history of the Rous sarcoma virus that runs through cancer research,” says Dr. Resh. “The src oncogene taught us how many basic cellular processes work, and also what goes awry in cancer.”
For that, we have a chicken to thank.