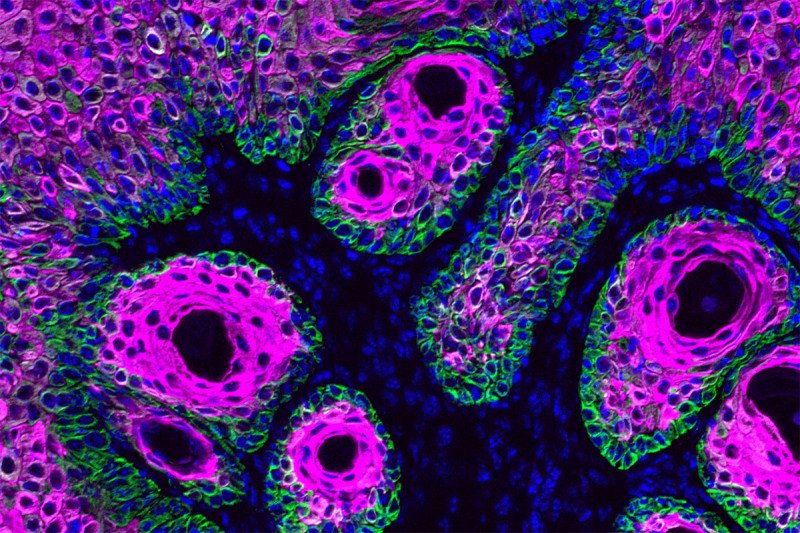
In recent years, cancer biologists have come to understand that metabolism — the way that cells acquire and use nutrients — can directly affect their tendency to become cancerous.
SKI cell biologist Lydia Finley and colleagues in the Elaine Fuchs lab at The Rockefeller University have now deepened knowledge of this relationship in the context of squamous cell carcinoma, a cancer that arises from stem cells in the skin. Using mouse models and cells growing in tissue culture, they found that the amount of the amino acid serine present in a stem cell’s environment influences its “decision” to keep dividing or to grow up (differentiate). Differentiated cells generally do not form cancer.
“The stem cells that give rise to squamous cell carcinoma seem to be highly dependent on extracellular serine for their growth,” Dr. Finley says. “Trying to ‘starve’ these cells of this source of serine could be a strategy to try to curb their growth by forcing them to differentiate.”
A normal stem cell will respond to a shortage of extracellular serine by synthesizing more. At the same time, they will begin differentiating: The biochemical pathways involved with serine synthesis interact with proteins called histones that wrap DNA like a spool of thread and allow specific genes to be turned on. Stem cells with cancer-predisposing mutations, on the other hand, seem intent on avoiding new serine synthesis.
Cancer stem cells’ heightened reliance on extracellular serine reflects what Dr. Finley calls “metabolic rewiring”: By relying on extracellular serine, the cancer stem cells can avoid serine synthesis, with the happy side effect (for the cancer cell) that the path toward differentiation is blocked.
“Our findings link the nutrients that a skin stem cell consumes to their identity and their ability to initiate a tumor,” says Sanjeethan Baksh, a Tri-Institutional MD/PhD student in the Fuchs lab and the paper’s first author. “Not only do nutrients allow stem cells and cancer cells to grow, but our study also shows that metabolism directly regulates gene expression programs important for cancer stem cell identity.”
Although restricting serine in the diet is not feasible in humans, the team is currently looking for ways that they might be able to interfere with cancer stem cells’ ability to take up serine in the hope of curbing cancer growth.
The findings were reported on May 25 in the journal Nature Cell Biology.
This study received financial support from the Howard Hughes Medical Institute, the National Institutes of Health (grants R01-AR31737, F31CA236465, F30CA236239-01, and 1F32AR073105), the Human Frontiers Science Program, the European Molecular Biology Organization, NYSTEM (CO29559), The Starr Foundation, the Damon Runyon Cancer Research Foundation, the Concern Foundation, the Anna Fuller Fund, The Edward Mallinckrodt, Jr. Foundation, and the Memorial Sloan Kettering Cancer Center Support Grant P30 CA008748. The study authors declare no competing interests.

