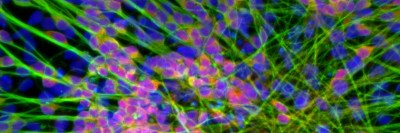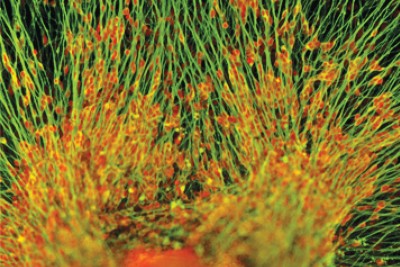
Trigeminal sensory neurons, nerve cells important for facial sensation and pain, that have been derived from human embryonic stem cells (Image provided by Zehra Dincer and Lorenz Studer)
To take advantage of the potential of embryonic stem cells and human adult cells to produce regenerative treatments and provide insights into cancer, the Sloan Kettering Institute has created a new Center for Stem Cell Biology (CSCB). Led by stem cell biologist Lorenz P. Studer, the virtual center will foster collaborations among Sloan Kettering Institute investigators working in the field.
“We’ve made important discoveries here, and this new center will allow us to capitalize on them,” Dr. Studer says. “It provides a critical mass of resources and expertise for the researchers already doing work at Memorial Sloan Kettering Cancer Center, and it will help us to continue bringing in top stem cell scientists in the future.”
The CSCB consists of a hub of several Sloan Kettering Institute laboratories, dedicated facilities, and administrative support. Scientists working under the umbrella of the CSCB will collaborate with other Memorial Sloan Kettering colleagues to develop novel technologies, recruit faculty, and provide stem cell training. Dr. Studer says the CSCB will complement Memorial Sloan Kettering’s Center for Cell Engineering (CCE), which was established in 2007 and is led by molecular geneticist Michel Sadelain. The CSCB will study the basic biology and mechanisms of stem cells, while the CCE will explore ways to bring stem cell therapies to the clinic.
Dr. Studer explains that the CSCB has four main areas of interest. One involves the mechanisms of stem cell self-renewal and pluripotency, which allow stem cells to continuously replicate while retaining their capacity to develop into various cell types. Another area focuses on learning how stem cells naturally differentiate into specialized cell types with the goal of mimicking this process in the laboratory.
A third area concentrates on the relationship between stem cells and cancer, particularly the study of stem-like cells within tumors, which may help explain resistance to therapy and the tendency of some cancers to return after treatment. Surgeon and researcher Viviane Tabar, a CSCB member, published an important finding about these stem-like cells in December 2010. (Read more about Viviane Tabar’s research.)
A fourth research area explores the use of induced pluripotent stem cells (iPSCs), a new kind of stem cell that can be produced when a human adult cell is reprogrammed to its original, pluripotent state. Scientists plan to learn how genetic diseases develop by converting patient cells to an iPSC state and then investigating how they first began to malfunction. In 2009, Drs. Studer, Sadelain, and Tabar, along with Gabsang Lee, a postdoctoral fellow in Dr. Studer’s laboratory, were the first to successfully model a human disease using iPSCs in a cell culture dish.
In addition to shedding light on disease, researchers also hope to eventually use iPSCs to generate new tissue that could be transplanted into patients. A cell taken from a patient would be converted to an iPSC and directed to develop into the type of tissue the patient needs. Because the replacement cells would be genetically identical to the patient, presumably there would be little risk of immune rejection.
Although Dr. Studer says it is too early to know if iPSCs can completely replace embryonic stem cells for purposes of research and therapy, CSCB members have already made important strides with iPSCs. In the January 2011 issue of Nature Biotechnology, Drs. Sadelain and Studer, along with Eirini Papapetrou, a postdoctoral fellow in Dr. Sadelain’s laboratory, and other Memorial Sloan Kettering colleagues, described a way to insert genes into iPSCs in genomic “safe harbors” — locations on the chromosomes that are far away from cancer-related genes — to avoid the risk of activating these genes. [PubMed Abstract] They used this method to correct beta thalassemia, a blood disorder caused by a deficit in hemoglobin production, using patient cells in culture.
“There are legitimate concerns about the safety of using pluripotent stem cells for therapeutic purposes,” Dr. Sadelain says. “We aim to identify sites in the human genome where genes can be safely inserted to correct or control the function of cells generated from patient-derived iPSCs. This strategy will allow us to develop improved therapies for genetic diseases, degenerative disorders, and cancer.”
The CSCB was publicly launched on November 16, 2010 with an inaugural symposium that featured presentations by four leading stem cell scientists from other institutions, each addressing one of the center’s four areas of interest, and a poster session with more than 40 stem cell-related presentations from New York City-area labs. The CSCB also was bolstered by training grants of approximately $1.86 million over five years from New York State, which will support five postdoctoral fellows in stem cell research beginning July 1, 2011.