Physician-scientist Samuel Bakhoum says, “Seeing trainees beaming over a new discovery is worth its weight in gold."
My life has taken a circuitous route. I was born in Nigeria and grew up in Alexandria, Egypt. I went to high school in Italy; college in Vancouver, Canada; then got my MD-PhD in New Hampshire at Dartmouth College. After a year in Boston doing a medical internship, I came to New York in 2014 for residency training at Memorial Sloan Kettering and research fellowship training at Weill Cornell. I’ve lived in many places, but I feel most at home here in New York City because it’s so international and cosmopolitan.
My interest in science started when I was a young child. I credit my dad because I asked him endless questions about how things are made and how they work. He was very patient and nurtured my curiosity. Both he and my mother are doctors. Up through high school, I assumed that being a doctor would include enough research to satisfy me. But as an undergraduate student, I began to realize that being a doctor and being a scientist are two separate paths. I temporarily pursued a science-only path, enrolling at Dartmouth as a graduate student in cancer biology. But I soon came to realize that combining science and medicine was my true calling.
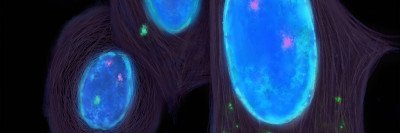
Around that time, I became interested in chromosomal instability, also called CIN. At any moment, millions of our cells are dividing. They must equally distribute their chromosomes. This is controlled by what is called the mitotic spindle. Sometimes the spindle can malfunction, and the distribution can go drastically wrong. Parts of chromosomes, or even entire chromosomes, end up outside the nucleus in micronuclei. Envelopes surrounding micronuclei often rupture and spill DNA into the cytoplasm, which is the liquid that surrounds the cell.
CIN and Cancer
We started to question whether CIN could cause cancer progression — and if it does, whether we can use genetic tools to reverse it. We wondered: Can we correct these mistakes? And can we also understand the role they may play in other cancer-related processes, such as when the disease spreads or becomes resistant to drugs? I found these questions very intriguing and continued studying CIN after I came to MSK for medical residency. During that time, I conducted research at Weill Cornell in the laboratory of Lewis Cantley.
In early 2018, Dr. Cantley and I published findings in Nature that DNA in the cytoplasm arising from CIN creates an inflammatory state within cells. In normal cells, inflammation either causes cells to self-destruct or activates an antiviral immune response that destroys the cells. Cancer cells have somehow evolved a way to withstand this type of destruction. Instead, they use the inflammation to their advantage. They hijack pathways used by immune cells to migrate so they can escape the primary tumor and spread to other parts of the body.
In September 2018, I set up my lab here at MSK in the Human Oncology and Pathogenesis Program (HOPP). Our primary research focus is gaining a better understanding of CIN and using this information for therapies that tackle aggressive cancers and their ability to spread. One emphasis is finding ways to suppress the instability in the first place. Another area is learning how to inhibit the inflammation. Theoretically, this could prevent the cancer cells from spreading.
A third strategy comes at the problem from the opposite direction. By creating chromosomal chaos, cancer cells are playing with fire. We might be able to further increase CIN or target certain vulnerabilities to tip the balance away from survival toward complete self-destruction.
A Research and Clinical Balance
My interest in CIN played a role in my choice of clinical work. A colleague at Dartmouth Medical School, who became a mentor, was a radiation oncologist. We were talking one day about how my research on CIN fits well with radiation research because radiation induces chromosomal damage. And even though at MSK most of us focus on one disease site — breast cancer in my case — as radiation oncologists, we’re trained to treat patients head to toe. This training provided me with a comprehensive understanding of how cancer is treated from beginning to end. I see people with breast cancer one day a week and work in the lab four days.
The environment in HOPP is one of the biggest benefits of being at MSK. No other institution has the unique structure centered on physician-scientists. The work in HOPP runs the gamut from basic science to translational and clinical research. We also have tremendous administrative support that lets us focus on research and mentoring young scientists. It’s a great place for people to be trained. My wife, whom I met a Dartmouth, came to MSK when I did and trained under Joan Massagué and Dana Pe’er. Later this year, she will be setting up her own lab at Weill Cornell across the street.
My favorite part of my job is seeing other people in my lab get excited about their own discoveries. I love it when they come into my office with a big smile about something they just found while analyzing the results of an experiment. Seeing trainees beaming over a new discovery is worth its weight in gold.