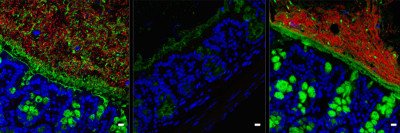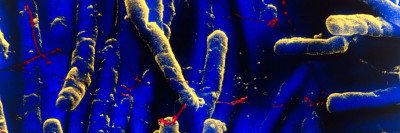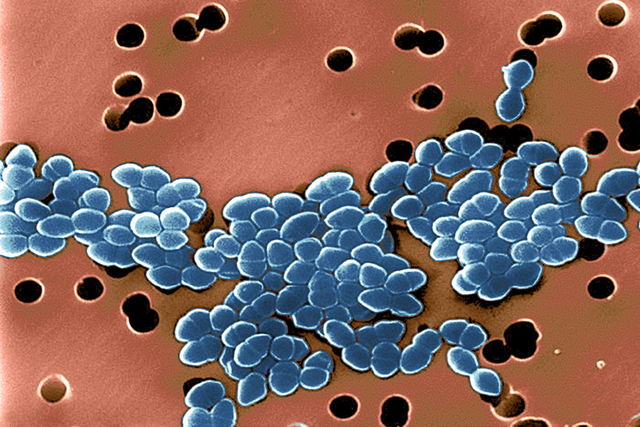
Hospital-acquired infections are a major threat, especially for people whose immune systems may be compromised because of cancer treatment. In recent years, researchers have been studying fecal microbiota transplants as a way to treat this serious complication. These transplants involve collecting stool from a healthy donor and delivering it into the intestine of the patient. The beneficial microorganisms from the transplant restore the balance of healthy bacteria in the gut.
Little is known, however, about which species of bacteria offer protection against harmful pathogens or exactly how they provide this benefit. A new study from Memorial Sloan Kettering is reporting the first evidence of a bacterial species that appears to maintain the balance of healthy microbes by killing dangerous ones. The findings also suggest how it mounts this attack. The research was reported August 21 in Nature.
“A lot of work is being done to figure out how harmful pathogens are able to colonize the human body,” says first author Sohn Kim, an MD-PhD student in the Tri-Institutional MD-PhD Program of MSK, Weill Cornell Medicine, and The Rockefeller University. “This project provides important new information about the bacteria that keep them in check.”
Focus on a Deadly Infection
The study focused on a particularly threatening hospital-acquired infection called vancomycin-resistant Enterococcus (VRE). VRE sickens about 20,000 people in the United States every year, according to the Centers for Disease Control and Prevention, and kills up to 10% of them. Earlier work led by former MSK graduate student Silvia Caballero, a co-author on the current study, showed that a mixture of four bacterial strains protect lab mice from VRE. These strains are normally found in the gastrointestinal tracts of healthy people.
The new study built on this earlier work by conducting a series of experiments to isolate one of these four bacterial strains: Blautia producta. “The next step was to determine the mechanism by which Blautia producta mediates protection against VRE,” Dr. Kim says. It turned out that a protein produced by Blautia producta is able to kill VRE even when the bacterial cells themselves aren’t present. Further study revealed that this protein is a lantibiotic, a type of antibiotic that is manufactured by microorganisms.
“If you think of Blautia producta as a member of the microbiota that helps maintain order within the gut, this lantibiotic is what it uses to do that,” says MSK infectious diseases expert Ying Taur, a co-author on the study. “This study really helps further our understanding of how all this works and provides important new insight.”
Evaluating the Effects of a Bacterial Product
The researchers did a number of additional studies. These included sequencing the gene that codes for the lantibiotic and performing RNA sequencing to determine when the gene is expressed.
They also tested the lantibiotic against about 150 strains of intestinal bacteria, to gain a sense of its spectrum of activity. This part of the research was significant because a major side effect of the antibiotics that doctors prescribe is that they can wipe out these healthy strains.
The team found that Blautia producta and the lantibiotic did not damage healthy strains. In fact, when they reviewed their library of samples collected from healthy donors, the researchers learned that about half of them already had Blautia producta and this lantibiotic product.
“It’s remarkable how precise this product is at targeting harmful microbes while sparing healthy ones,” Dr. Taur notes. “This is something we do not know how to do with any antibiotics that we have now. Our antibiotics are very clunky in comparison to the precision of what these bacteria do.”
Moving Forward with More Research
More work is needed before this approach can be tested in people with VRE infections. Drs. Kim and Taur say they haven’t even determined how a treatment would be best administered or whether they would use Blautia producta or the isolated lantibiotic. The treatment could possibly be given as a pill, or the findings from this study could be used to develop a more specialized type of fecal microbiota transplant. They plan to study various approaches in mouse models.
“Previously, studies have shown that Blautia is associated with better outcomes in people who have developed graft-versus-host disease (GVHD) after having a bone marrow transplant with donor cells,” says study co-author Marcel van den Brink. “In addition, we have recently found that Enterococcus is associated with increased incidence of GVHD. These findings offer exciting opportunities to control GVHD and improve outcomes for people having transplants.”
“There are a lot of things we still don’t know, but we have learned so much from this study,” Dr. Taur concludes. “It was really an amazing piece of detective work.”
This research was led by former MSK physician-scientist Eric Pamer, who recently joined University of Chicago Medicine. It was funded by National Institutes of Health grants (RO1 AI42135, RO1 AI95706, UO1 AI124275, and P30 CA008748); the Lucille Castori Center for Microbes, Inflammation, and Cancer at MSK; a Medical Scientist Training Program grant from the National Institute of General Medical Sciences; an Early Postdoc Mobility fellowship from the Swiss National Science Foundation; and an Irvington Postdoctoral Fellowship from the Cancer Research Institute.
Dr. van den Brink is on the advisory board of and has financial holdings in Seres Therapeutics, serves on the DKMS medical council, has received speaker fees from Merck and the Acute Leukemia Forum, holds patents that receive royalties from Seres Therapeutics, has received fees and research support from Seres Therapeutics, and has intellectual property licensing with Seres Therapeutics and Juno Therapeutics.