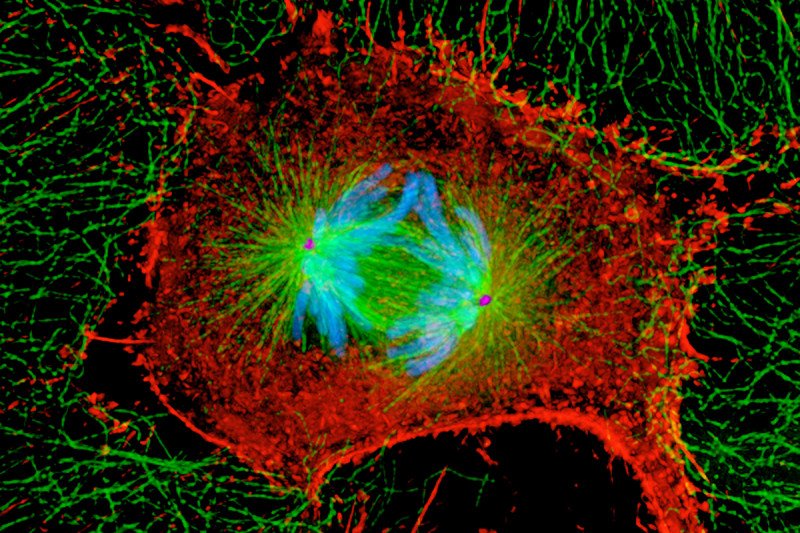
To create functioning tissues and organs, cells have to divide with precision. During one of the most important events in cell division, called mitosis, a cell splits into two identical copies of itself, called daughter cells. However this process doesn’t always go smoothly. Errors can occur, which can cause a cell to become cancerous.
A new study, recently published in the journal Developmental Cell, uncovered important new details about one of the key ways a dividing cell is able to recognize and correct these mistakes. Prasad Jallepalli, a researcher in the Sloan Kettering Institute’s Molecular Biology Program, led the research effort.
“The process of making, detecting, and correcting errors happens all the time in cell division,” Dr. Jallepalli says. “What my lab is focused on is understanding the cell’s core mechanisms for quality control during mitosis.”
Fishing for Answers
Before mitosis can occur, the mother cell must duplicate and collate its DNA into rod-shaped structures called chromosomes. The twin DNA copies in each chromosome are called sister chromatids. As the cell divides into two, the sister chromatids are pulled apart. One set goes to each daughter cell.
“Imagine the sister chromatids are pairs of fish that are being hooked by fisherman on opposite shores, with the goal of having one of every pair in each fisherman’s catch,” Dr. Jallepalli explains. “Occasionally one fisherman’s hook may get attached to the wrong fish. When that happens, it needs to be fixed.
“What we’ve discovered is a new process by which the cell can recognize that the wrong connection was made,” he adds. “Then the cell can stop dividing until it unhooks the fish and reattaches the hook in the proper place, correcting the problem.”
In this analogy, the fishing lines are microtubules. These tiny thread-like structures control the movements of the many parts within a cell, including helping to pull apart the chromosomes during mitosis. The hook is a protein called Ska. It attaches the chromosomes to the microtubules. In this study, the researchers reported for the first time how Ska is able to transform its activity and let go of the microtubule after the cell realizes that a mistake has been made.
Focusing on Kinases
Dr. Jallepalli says it’s important to study this mechanism to gain a fundamental understanding of the inner workings of our cells. Properly separating chromosomes is important for making sure that cells have all the genetic instructions they need to carry out their functions.
But this research also has wider implications for developing better, more targeted cancer therapies. A growing number of targeted cancer drugs act on proteins called kinases, which are enzymes that drive cellular activities by adding a chemical mark called phosphorylation. This study identified an important kinase that is involved in chromosome segregation. Eventually researchers may be able to develop therapies that disrupt this process, or others, in tumors.
A New Way to Investigate Kinase-Targeted Drugs
“The way we got to these findings represents a new paradigm for studying kinases,” Dr. Jallepalli says. “By using a burgeoning technique called quantitative proteomics, we were able to find dozens of proteins that were chemically modified by one kinase, out of a soup of tens of thousands of proteins in the cell, many of which are modified by other kinases.” Quantitative proteomics is a technique of analytical chemistry. It allows researchers to obtain detailed information about the levels and chemical state of proteins within different samples.
“These types of insights were unimaginable ten years ago,” he concludes. “It’s a testament to how quickly this technology has evolved, and it gives us a new way of studying kinase-driven biology and developing drugs that target kinases involved in disease.”
This research was supported by the National Institutes of Health (grants R01GM094972 and P30CA008748), a Wellcome Trust Senior Investigator Award, and a Royal Society Wolfson Research Merit Award.