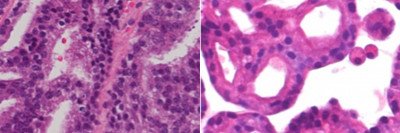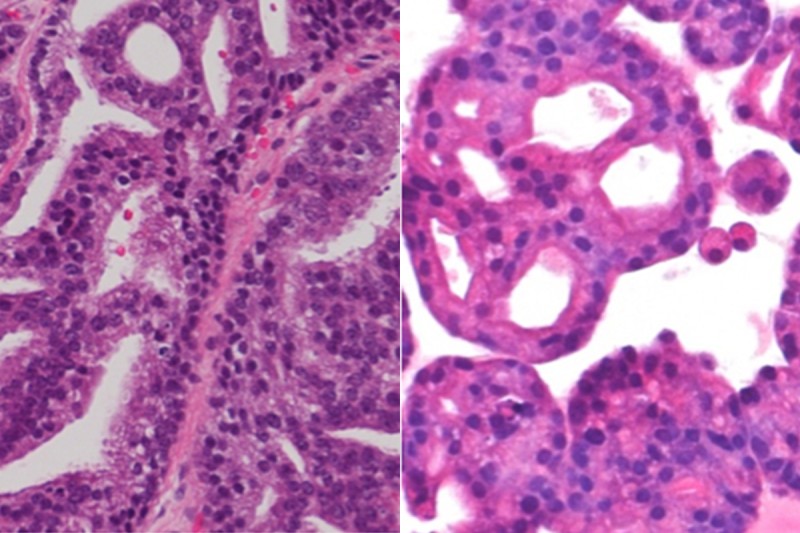
Stained pathology slides of a patient’s tumor (right) and of an organoid made from that tumor (left). The development of these cancer organoids is one of the many innovations that MSK researchers studied in 2014.
This has been a busy year for scientists at Memorial Sloan Kettering. Our dedicated researchers have worked ceaselessly on a huge variety of approaches to advance cancer science and to bring those innovations into practice for patients as quickly as possible. Here’s a snapshot of some of the fascinating work they did in the last 12 months.
Making Cancer Cells Normal
Conventional cancer treatment involves getting rid of cancer cells through surgery, radiation, or drug therapy. But what if there were a way to change cancer cells back into normal ones instead of killing them? That’s exactly the approach hematologist Eytan Stein and colleagues took to treating certain types of leukemia and other blood diseases such as myelodysplastic syndromes. A drug they’re pursuing blocks the mutant enzymes that prevent healthy bone marrow cells from developing. “This approach is truly transformative,” Dr. Stein said. “Both because it changes the way we look at cancer and because it literally transforms cells.”
Leaving No Cancer Cells Behind
In the operating room, radiologist Michelle Bradbury led work to use an innovative optical nanoparticle probe in an effort to ensure that no stray cancer cells remain after surgery. The nanoparticles, which are loaded with fluorescent dye as well as a molecule that causes them to bind to the surface of tumor cells, are injected during the operation to remove the cancer. Then doctors use a special handheld camera that causes the fluorescent dye to light up the nanoparticles and show where they’ve traveled. “We are trying to push the optical properties and brightness of the particles to the point that we could detect even a single cancer cell,” said Dr. Bradbury. The technology is currently being used in patients with melanoma of the head and neck. Watch a video about the probe.
Creating New Cancer Models
Medical oncologist Yu Chen explored a promising new tool called an organoid for testing the efficacy of cancer treatments. The study involved isolating tumor cells taken from seven patients with metastatic prostate cancer and then growing them in the laboratory. Each of the resulting organoids was molecularly identical to the patient’s cancer from which it originated. Eventually, Dr. Chen explained, they may be able to develop organoids derived from every patient’s tumor, allowing doctors to test a potential therapy on the organoid before giving it to the patient for a truly personalized and optimized approach to treatment. Read the full story.
Devising a Nematode Playbook
How can worm development contribute to cancer research? Computational biologist Zhirong Bao and his team studied a worm called a nematode to answer that question. Its simple anatomy and pattern of development make it the perfect model for testing new imaging tools created in Dr. Bao’s lab that track the development of cells and their genes in a series of high-definition 3-D snapshots. The resulting image data provided the information necessary to complete a veritable biological playbook detailing how a particular tissue or organ develops. Ultimately Dr. Bao hopes the technology can be used to speed the study of the intricate cellular interactions that cause tumors to grow and spread. Watch a nematode develop.
Growing Plant Therapy
For nearly 40 years, MYC has been one of the most well-studied yet perplexing of the so-called oncogenes — that is until this year, when a team of MSK researchers discovered that a compound found in the tropical plant Aglaia foveolata could block MYC’s cancer-causing activity. The compound, Silvestrol, destroyed leukemia and lymphoma cells as well as some solid tumors in animal models. Silvestrol causes cancer cells to die because the cells are dependent on the proteins encoded by MYC. “Blocking the production of key cancer genes is a completely new way of treating cancer. That is exciting, and it also means we have a lot to learn,” said senior author Hans Guido-Wendel. Read the full story.