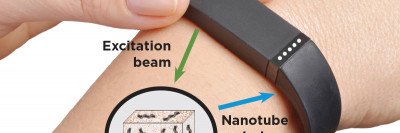
Imagine a device like an Apple Watch that could detect cancer in its earliest stages. The device would communicate with a tiny nanosensor, implanted in your body, that screens for the presence of cancer-associated markers. If any are detected, the sensor sends a signal to the watch, alerting you and your doctor.
This may sound like science fiction, but Sloan Kettering Institute biomedical engineer and Head of the Cancer Nanomedicine Laboratory Daniel Heller is working hard to make this vision a reality.
“For many cancers, if you can detect the disease early, then your chances of being cured are going to be a whole lot better,” Dr. Heller says. “That’s why we are so focused on developing nanosensors for early detection.”
The nanosensor technology is especially relevant to ovarian cancer, which is a major killer of women. Ovarian cancer causes few symptoms as it’s developing and there is no effective screening test, which is why it’s usually diagnosed at later stages. By that point, the disease has spread to areas outside of the ovary and is much more difficult to cure.
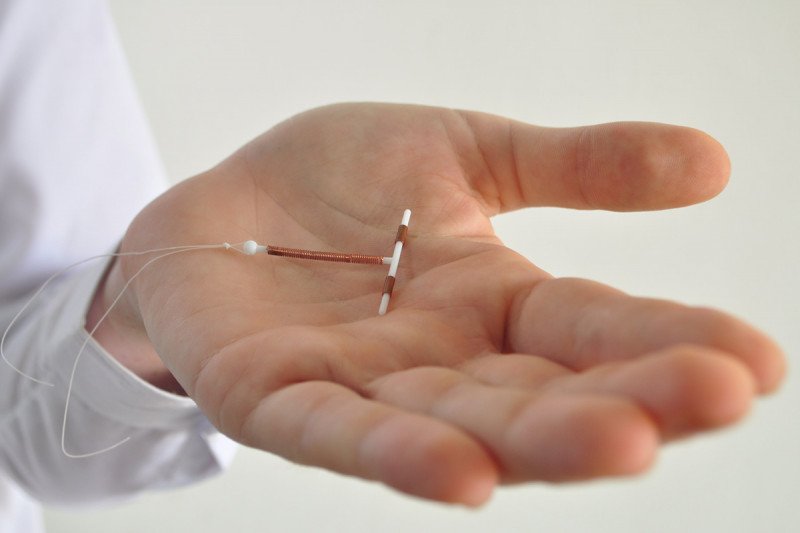
Dr. Heller and his team are developing several different types of nanosensors to detect ovarian cancer. One is designed to be implanted directly into the uterus, like an intrauterine device (IUD). Other nanosensors are designed to detect cancer-associated proteins in the blood, including one that can be implanted right under the skin.
The ultimate goal is to offer the technology to people at higher-than-average risk of ovarian cancer — for example, those with inherited BRCA mutations.
How the Nanosensors Work
The sensors take advantage of the fact that ovarian cancer cells produce and release distinctive proteins into their surroundings. Some of these proteins enter the bloodstream, but the amounts are minute and hard to detect with standard blood tests. A sensor placed close to the source of the proteins (or biomarkers) — say in the fallopian tubes or in the uterus, where the concentration of these molecules is higher — could in theory find cancer early.
To detect these scarce biomarkers, Dr. Heller’s team is relying on something called carbon nanotubes. These tiny, rod-like particles are about one nanometer thick, which is about 1/100,000th the thickness of a human hair. The nanotubes emit harmless infrared light that can be picked up by a detector worn outside the body. The nanotubes are attached to another molecule called an antibody that recognizes and binds to a specific cancer biomarker.
“This binding event causes the light given off by the nanotube to change color, which we can detect even outside of the body,” says Zvi Yaari, a research scholar who leads the project in the Heller lab, working in collaboration with research fellow Ron Feiner and MSK gynecologic surgeon Kara C. Long, MD.
Previous work from the Heller lab has shown that an implantable nanosensor can detect ovarian cancer in mice, based on the presence of a biomarker called HE4. They are now developing one to detect the most common ovarian cancer biomarker, CA125.
“The greatest challenge we face in taking care of patients with ovarian cancer is tackling widespread disease,” Dr. Long says. “A tool that can find disease in its early stages, or possibly even in the pre-cancer stages, would save lives. This could be especially impactful for patients at increased risk for ovarian cancer, such as those with genetic mutations or family history of the disease.”
With the help of Dr. Long, Dr. Heller and his team are conducting their first tests in human tissues. They are implanting the nanosensors into uteri that have been removed during surgery.
“This will allow us to test how these sensors work before even putting them in humans,” Dr. Heller says.
Disease Fingerprinting
Another type of nanosensor Dr. Heller’s team is developing takes a more kitchen-sink approach: Rather than measuring individual protein biomarkers, this version detects thousands of potential markers all at once using many different nanosensors. It then uses machine learning to find associations between specific patterns of sensor responses and specific disease states — “like a disease fingerprint,” says Mijin Kim, a postdoctoral fellow in the lab who leads this project, in collaboration clinical chemist Lakshmi Ramanathan and chemists and engineers at Lehigh University, the University of Maryland, and the National Institute of Standards and Technology.
The sensor technology can thus far identify ovarian cancer more accurately than the current clinical screening test for biomarker CA125. One day, it may enable rapid and inexpensive screening.
“It would be great to be able to be monitor for disease progression without endless blood tests,” Dr. Kim says. “Can we at least lengthen the time in between doctor visits? That would be a big improvement.” For this work, Dr. Kim was awarded the 2021 Kravis WiSE Postdoctoral Fellowship. A paper discussing this work in more detail was published March 17, 2022 in the journal Nature Biomedical Engineering.
High Demand
Worldwide, more than 238,000 women are diagnosed with ovarian cancer each year, and more than 151,000 people die of the disease. Ovarian cancer is the fifth leading cause of cancer-related deaths among women in the United States, and the first among gynecological cancers.
Overall, the five-year survival rate is 46%. However, when ovarian cancer is diagnosed at stage I, the five-year survival rate is 92%.
The great need and great potential to improve cancer care for women is a main reason why Dr. Heller and his team are focusing their efforts on ovarian cancer first.
“We’re motivated by the prospect of truly making a big difference in people’s lives with this technology,” he says.
- There is currently no way to detect ovarian cancer early on, which is one of the reasons it is so difficult to cure.
- Nanosensors may aid the early detection of ovarian cancer by measuring microscopic amounts of cancer-associated proteins that are emitted by the tumors.
- The nanosensors are composed of extremely small carbon nanotubes that bind to cancer proteins and emit a distinct color of light, which can be picked up by a detector.
This work was supported in part by the Cancer Center Support Grant (P30-CA008748), the National Science Foundation CAREER Award (1752506), the Honorable Tina Brozman Foundation for Ovarian Cancer Research, the Tina Brozman Ovarian Cancer Research Consortium 2.0, the Kelly Auletta Fund for Ovarian Cancer Research, the Commonwealth Foundation for Cancer Research, the American Cancer Society Research Scholar Grant (GC230452), the Pershing Square Sohn Cancer Research Alliance, the Expect Miracles Foundation – Financial Services Against Cancer, and MSK’s Cycle for Survival Equinox Innovation Award for Rare Cancers. Dr. Yaari was supported by the Ann Schreiber Mentored Investigator Award (Ovarian Cancer Research Fund) and Young Investigator 2019 (Kaleidoscope of Hope). Dr. Kim was supported by the Marie-Josée Kravis Women in Science Endeavor Postdoctoral Fellowship. Dr. Heller is a cofounder and officer with equity interest of Goldilocks Therapeutics Inc., LipidSense Inc., and Nirova Biosense Inc., as well as a member of the scientific advisory board of Concarlo Holdings LLC, Nanorobotics Inc., and Mediphage Bioceuticals, Inc.