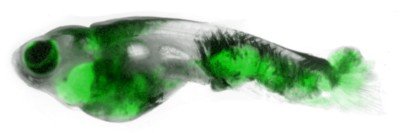
Cancer cells get hungry. Very hungry.
It takes a lot of energy to reproduce the way they do — repeatedly and often. Pound for pound, fat has more energy than any other nutrient. So it’s perhaps not surprising that when cancer cells find themselves in fat tissue, they make quick use of these resources.
According to a new study from researchers at the Sloan Kettering Institute at Memorial Sloan Kettering, the presence of fat may even be what helps cancer cells take root in the first place. Richard White, a physician-scientist in the Cancer Biology and Genetics Program at SKI, discovered that melanomas preferentially grow near adipose (fat) tissue and eagerly eat up the abundant fats, also called lipids, found there.
“This is the seed-and-soil hypothesis,” says Dr. White. “Tumor cells like to go to places where there is fertile soil. Based on the results of our study, we think that adipose tissue can be very fertile soil for melanoma.”
Knowing that adipose tissue enables some cancer cells to grow and spread suggests that cutting off their fat supply could be a way to fight the disease.
The findings also add to the growing understanding of the link between obesity and cancer.
Follow the Fat
Dr. White and his colleagues did not start out looking to implicate fat in cancer. They stumbled upon the connection.
His team uses zebrafish as a model system for studying skin cancer. These small freshwater fish get melanomas that are very similar to human melanomas. The fish are also transparent, which makes it easy to see where cancer cells go as tumors progress. The team was interested in identifying factors that might influence cancer’s ability to spread.
“We did a screen to look for what helped melanoma cells grow in certain locations,” Dr. White says. “When we looked at what was different in the melanoma cells growing at metastatic sites, we found a lot of changes in genes that regulate how cells use lipids.”
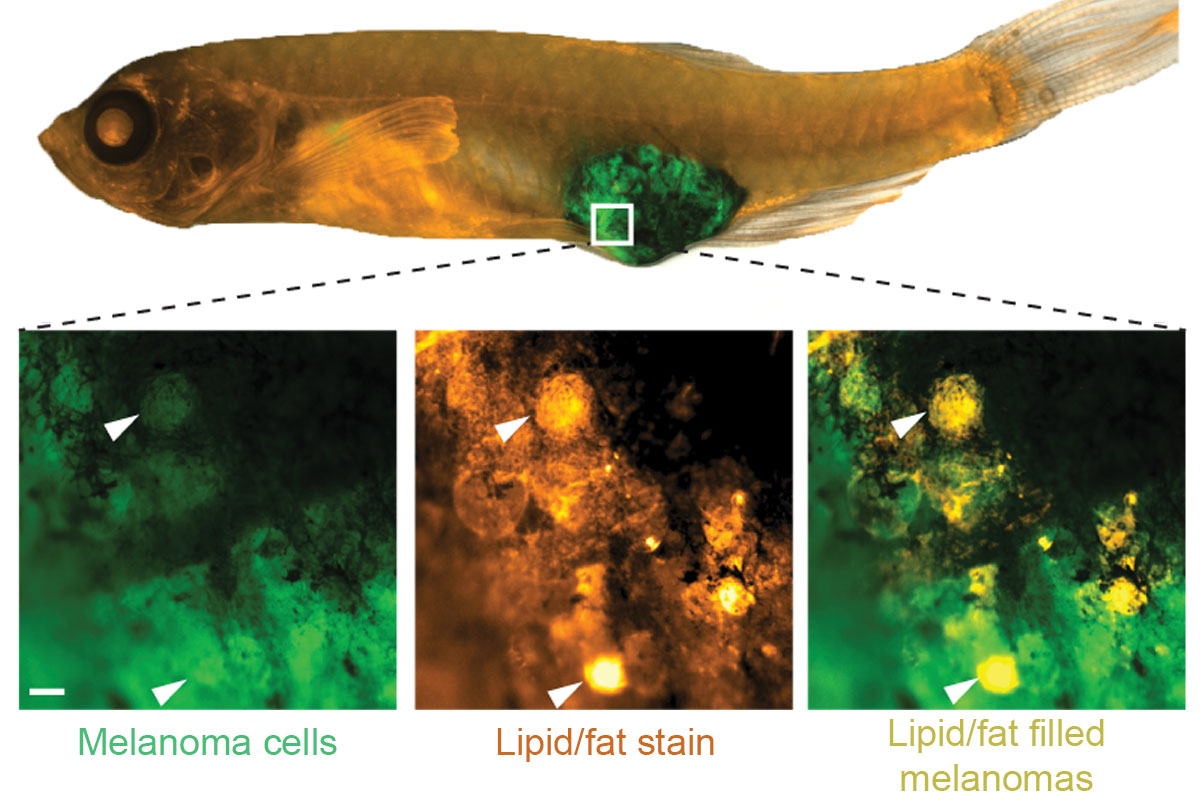
To investigate, postdoctoral fellow Maomao Zhang labeled the lipids in the adipocytes with a fluorescent marker. Then she put the adipocytes and melanoma cells together in a dish and followed where they went. The melanoma cells sucked up the lipids.
But that was in a dish. What about in living animals?
First they turned to their fish. Dr. Zhang injected melanoma cells next to adipocytes in fish. Sure enough, the melanoma cells accumulated lipids. Moreover, as these melanoma cells grew away from the injection site to form metastases, more than half of these tumors were found growing near adipocytes.
Next, the researchers examined the situation in people. They looked at tumor samples from people with melanoma who were treated at MSK. Just like in the fish, the human cancer cells also contained deposits of fat.
Rich Soil for Growth of Cancer
Melanoma cells that take up fat proliferate more and become more aggressive than those that don’t, the team discovered. The fat-fueled cells can chew through collagen and cross membranes with ease, allowing them to spread. They also change their metabolism, preferring to burn fat for energy instead of sugar.
This led the team to wonder whether blocking the cancer cells’ ability to take up fat would dampen their aggressiveness. They used a drug to block a protein, called the FATP transporter, that enables the cancer cells to take in fat. Cancer cells have a lot more of this transporter than normal cells, so they are more sensitive to the drug. As predicted, reducing the cells’ ability to take up fat slowed their growth and spread.
These results, Dr. White says, raise the possibility that fat-blocking drugs could open up a new treatment approach for melanoma in people.
“Can we identify a subset of patients whose tumors are particularly lipid dependent? Those might be the ones who could benefit from an approach geared toward blocking fat uptake into the cells,” he says.
Obesity and Cancer
An obvious question raised by these findings is whether they help to explain why obesity is such a strong risk factor for cancer. While his study doesn’t directly address the matter, Dr. White says it does show a concrete link between lipids and cancer growth. In that sense, it adds one piece to what is likely a very large puzzle.
Whether altering one’s diet to consume less fats might forestall melanoma progression is another big open question, one the team is interested in exploring further.
The study shows the utility of model systems like zebrafish for studying cancer, and the advantage of a place like MSK, where basic scientists and clinicians can easily collaborate.
“We started this in the zebrafish and were able to take it all the way through to human tissues,” Dr. White says. “It would have been tough to pull this off at another institution.” Among those MSK melanoma specialists who collaborated on the project were Jedd Wolchok, Charlotte Ariyan, Travis Hollmann and Paul Chapman.
A paper describing these results appears today in the journal Cancer Discovery.
This work was supported by the National Institutes of Health Director’s New Innovator Award (DP2CA186572), the Mentored Clinical Scientist Research Career Development Award (K08AR055368), the Melanoma Research Alliance, the Pershing Square Sohn Foundation, the Alan and Sandra Gerry Metastasis Research Initiative at Memorial Sloan Kettering, the Harry J. Lloyd Foundation, Consano, the Starr Cancer Consortium, the Memorial Sloan Kettering Translational Research Oncology Training Program fellowship, and National Cancer Institute F32 Postdoctoral Training Grant (CA210536-01A1).