Cancer Biology & Genetics Program
The Robert Benezra Lab
Research

Professor
The overarching goal of the lab is to understand molecular mechanisms that contribute to cancer development and progression. My laboratory has had a longstanding interest in the role of the ID proteins (particularly ID1 and ID3) in normal development and cancer biology. ID proteins block tissue specific gene expression associated with changes in cell state and their expression is normally confined to early embryogenesis to block differentiation but are reactivated in a variety of cancer types. We have contributed to the development and distribution of reagents and animal models related to ID proteins that have proven to be valuable in establishing the role of these proteins in promoting cancer and other pathologic states as well as their role in normal stem cell biology. Genetic analyses of ID loss-of-function in cancer have proven their importance in disease initiation and progression which motivated us to develop a small molecule antagonist of the ID protein family that leads to ID degradation is being readied for clinical application. Both genetic analyses and drug treatment in animal models points to a requirement of ID proteins for mesenchymal-to-epithelial transition required for the formation of macro-metastases.
We have also found recently that ID proteins are essential for the development of non-alcoholic steatohepatitis (NASH) by controlling a gene regulatory pathway in a subpopulation of liver cells. NASH, highly prevalent in populations that consume high fat diets, is a significant risk factor for the development of liver cancer so the use of our ID antagonist may have important utility in cancer prevention. We also helped initiate the biochemical and cell biological characterization of the metazoan mitotic checkpoint pathway through the cloning and characterization of hsMad2, a key component of the pathway which blocks sister chromatid separation until all kinetochores are occupied by microtubules. In addition, we helped lead the efforts to understand the consequences of deregulated expression of Mad2 in the genomic instability commonly observed in cancer.
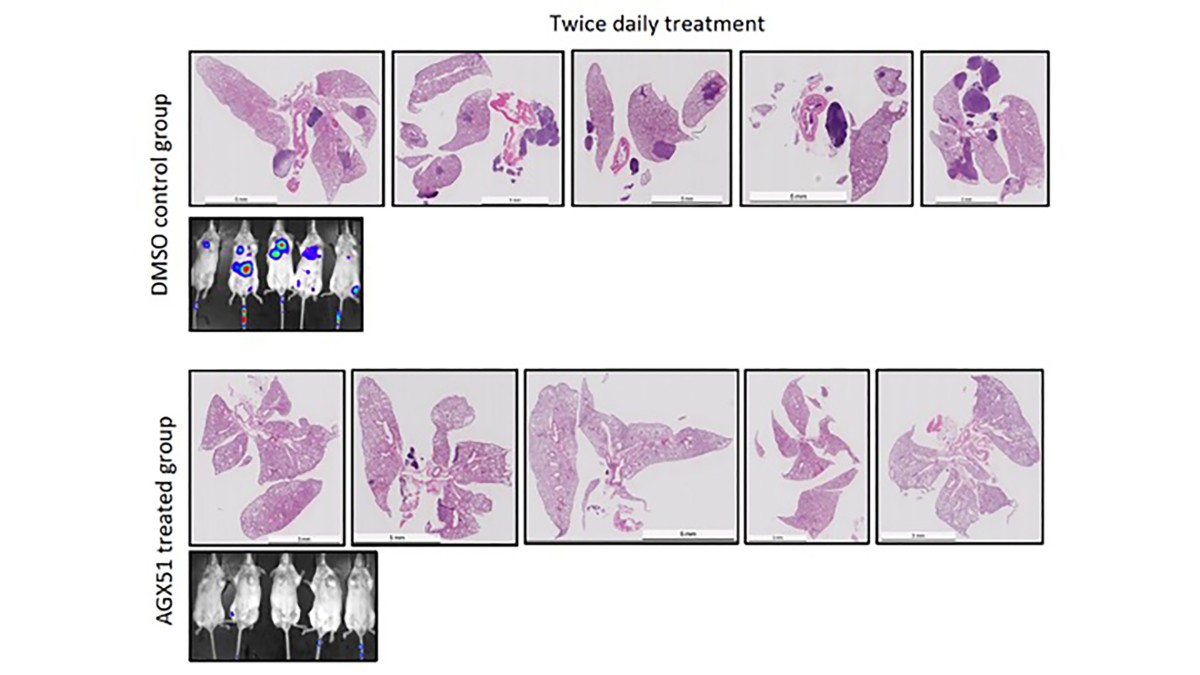
Inhibition of metastasis by AGX51, an Id degrader
Featured News
Publications Highlights
Benezra, R., Davis, R.L., Lockshon, D., Turner, D.L. & Weintraub, H. (1990). The protein Id: a negative regulator of helix-loop-helix DNA binding proteins. Cell 61(1):49-59.
Jen, Y., Weintraub, H. & Benezra, R. (1992). Overexpression of Id protein inhibits the muscle differentiation program: in vivo association of Id with E2A proteins. Genes Dev 6(8):1466-79.
Pesce, S. & Benezra, R. (1993). The loop region of the helix-loop-helix protein Id1 is critical for its dominant negative activity. Mol Cell Biol 13(12):7874-80.
Tournay, O. & Benezra, R. (1996). Transcription of the dominant-negative helix-loop-helix protein Id1 is regulated by a protein complex containing the immediate-early response gene Egr-1. Mol Cell Biol 16(5):2418-30.
Li ,Y. & Benezra, R. (1996). Identification of a human mitotic checkpoint gene: hsMAD2. Science 274(5285):246-8.
People

Robert Benezra, PhD
Deputy Director for Core Technologies; Laura and Christopher Pucillo Chair in Metastasis Research
Professor
- Cancer biologist Robert Benezra studies the molecular mechanisms of tumor growth and metastatic progression using mouse models. In addition, he studies the spindle assembly checkpoint which ensures proper chromosome segregation and is often deregulated in cancer.
- PhD, Columbia University
- [email protected]
- Email Address
- 646-888-2812
- Office Phone
Members

- 646-888-2813/2814
- Lab Phone

Lab Head; Laura and Christopher Pucillo Chair in Metastasis Research

Research Assistant

Research Assistant

Research Fellow

Research Fellow







Lab Alumni

Graduate Student

Research Fellow

Research Technician

Research Fellow


Assistant to Dr. Robert Benezra

Graduate Student

Graduate Student

Graduate Student

Graduate Student

Graduate Student

Research Assistant

Graduate Student

Graduate Student

Research Fellow

MD-PhD Student

Research Fellow

Research Fellow

Research Fellow

Graduate Student
Get in Touch
-
Lab Head Email
-
Office Phone
-
Lab Phone
-
Lab Fax