If you have ever been to a professional baseball game and sat close to the field, you may have experienced the thrill of watching a player scoop up a ball in right field and fire it to third base. As the ball travels 100 yards in less than two seconds and the teammate makes the tag, you might think to yourself, how can anyone throw a ball that fast, with such power and accuracy?
The answer is muscles — finely trained and operating at peak performance.
Muscles do many amazing things for us, from powering our hearts and churning our stomachs to keeping us walking for a lifetime. When muscles fail us, the results can be disastrous.
What accounts for muscles’ great power and versatility? It comes down to their unique structure.
Unlike a skin cell, say, which is small, flat, and round, a muscle cell is long, thin, and ropelike. Muscle fibers can slide past each other, pulling and releasing to generate movement.
Also, whereas a skin cell has a single command center — its nucleus — a muscle cell has many. They need these multiple command centers to send marching orders throughout the entire length of the cell. But this structure raises some fundamental questions.
“In a typical cell that has only one nucleus, everybody looks to the nucleus and says, ’You’re the brain of the cell. You’re in charge of transcription and translation,’” says Mary Baylies, a developmental biologist in the Sloan Kettering Institute. “But in a muscle cell, there are multiple brains. So how do they decide to give a muscle cell a certain size, shape, and physiology? Biologists haven’t really addressed that question.”
In a paper published on March 21 in the journal Developmental Cell, she and her colleagues provide the first evidence for a kind of communal decision-making that collectively determines muscle cell size.
The findings will be of interest to researchers and clinicians who study muscle diseases — and to cancer doctors, too, as muscle atrophy, or wasting, contributes to more than 50% of cancer deaths.
The Flies Have It
In the field of muscle cell research, the traditional view is that each nucleus in a muscle cell supplies only its immediate surrounding area with the gene products (proteins) it needs. In that sense, the area surrounding each nucleus in a muscle cell operates as a kind of fiefdom.
But that hypothesis only raises more questions, says Stefanie Windner, a postdoctoral fellow in the Baylies lab and the first author on the new paper: “Do those nuclei cooperate? Do they have their own strict program? Do they talk to each other?”
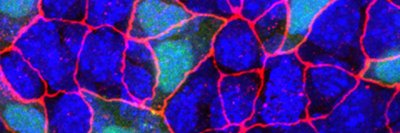
Dr. Windner sought to answer these questions using a unique model system: the abdominal muscles of a fruit fly larva. Flies have 60 abdominal muscles in each segment of their body. Each muscle consists of a single cell with multiple nuclei. This one-cell-per-muscle arrangement is useful for conducting experiments.
Using this system, she was able to systematically alter elements of a muscle cell’s components and see how the cell as a whole responded. For example, she changed the amount of DNA in nuclei and noted whether the size of the nucleus changed (not always). She asked what effect there was if the nuclei were near a nerve junction (more DNA content). She looked at whether physical force, like tugging, affect the position of nuclei (they do).
After recording all of these measurements, Drs. Windner and Baylies then worked with mathematicians Angelika Manhart and Alexander Mogilner at New York University to devise an equation for muscle cell size that factors in all of these parameters.
“In a sense, we can view the nuclei in muscle cells as players on a team, with each doing its part to keep runners off the bases,” says Dr. Baylies. “Some nuclei are like the pitchers and are busy keeping the batters from hitting the ball. Others are busy seeing that hits don’t turn into runs. But a system of rules connects them, so they aren’t operating as independently as it might seem.”
Dr. Baylies and her team are now looking for the rule book that explains how the nuclei know how to cooperate and plan together, without any single one being in charge.
Some of the questions the researchers plan to ask: How do the nuclei within the cell communicate? How do they set up differences between them yet effectively coordinate their activity? What are the duties of the different nuclei? How do these conditions change with disease, like cancer, or during aging?
Though challenges remain, Dr. Baylies is hopeful about the prospects for success. “With the right questions and techniques, we will get there,” she says.